Diagnosed with Cancer? Your two greatest challenges are understanding cancer and understanding possible side effects from chemo and radiation. Knowledge is Power!
Learn about conventional, complementary, and integrative therapies.
Dealing with treatment side effects? Learn about evidence-based therapies to alleviate your symptoms.
Click the orange button to the right to learn more.
- You are here:
- Home »
- Blog »
- side effects ID and prevention »
- 25% Chemo Cardio-toxic- Heart Failure?
25% Chemo Cardio-toxic- Heart Failure?
According to the article linked below, the FDA warns that 1 in 4 chemotherapy regimens cause heart damage. But don’t worry. The risk of heart failure is quite small.
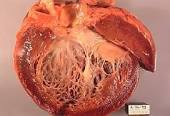
I was diagnosed with multiple myeloma in early 1994. I underwent VAD induction therapy and an autologous stem cell transplant in 12/95. I developed heart failure 15 years after my stem cell transplant.
I developed chronic atrial fibrillation (Afib) in December of 2010 and chemotherapy-induced cardiomyopathy. The issue of cardiotoxic chemotherapy regimens is near and dear to my heart.
First and foremost, conventional oncology doesn’t really know what the risk of late stage heart failure is. Secondly, that’s what people say until the problem happens to them. Risk is always small until it happens to you…
Further, I’m not saying that cancer patients should “stop using cancer drugs that save lives…” Not at all. I am saying two things.
- First, I am saying that a host of evidence-based complementary therapies have been shown to reduce the risk of heart failure from cardio-toxic therapies. Oncologists should prescribe frequent, moderate exercise, heart healthy nutrition, supplementation such as CoQ10, etc. to reduce the risk of chemotherapy-induced heart failure.
- Secondly, I am saying that oncologists should NOT WAIT for a cancer patient to start “showing signs or symptoms of a potential heart issue” as Dr. Daniel Addison, MD, advises before they do something. I didn’t exhibit any signs of “a potential heart issue“ during my five cycles of VAD induction chemotherapy. I woke up one December morning and flipped into chronic/permanent atrial fibrillation (Afib). This happened fifteen (15) years after VAD therapy.
How many other cancer survivors who have undergone cardio-toxic chemo who’s hearts have been damaged yet are not yet exhibiting signs of heart failure!?!? How many cancer survivors are walking around with:
- Chemotherapy-induced Cardiomyopathy (read more 1)
- Chemotherapy-induced Atrial Fibrillation (read more 2)
- Chemotherapy-induced Hypertension (read more 3)
- Chemotherapy-induced Valvular Damage (read more 4)
- Chemotherapy-induced Thrombosis (read more 5)
The fact that FDA warnings are issued years after formally approving the drug tells me that evidence of heart damage often occurs long after the patient completes his/her active therapy.
If you are currently undergoing one of the many known cardio-toxic chemotherapy drugs, I encourage you to also undergo evidence-based, non-toxic, heart-healthy complementary therapies such as nutrition, moderate exercise, supplementation, and lifestyle therapies.
To learn more about heart healthy complementary therapies, scroll down the page, post a question or comment and I will reply to you ASAP.
Thank you,
David Emerson
- Cancer Survivor
- Cancer Coach
- Director PeopleBeatingCancer
Recommended Reading:
- Chemotherapy-Induced Aging- Non-Toxic Therapies
- Managing Cancer- Chemotherapy, Radiation, Antioxidants
FDA Issued Postmarket CVD Safety Warning for 1 in 4 Cancer Drugs
“The US Food and Drug Administration (FDA) has issued postmarket cardiac safety warnings for about 1 in 4 cancer therapies, according to a research letter published September 30 in JAMA Oncology…
The US Food and Drug Administration (FDA) has issued postmarket cardiac safety warnings for about 1 in 4 cancer therapies, according to a research letter published September 30 in JAMA Oncology.
These cardiotoxic risk communications typically were issued just over 4.5 years after a drug’s approval — 40% longer than it took to issue noncardiac warnings.
However, lead author Daniel Addison, MD, explained that the likelihood of having a major cardiac event related to a cancer drug appears to be quite small.
“I don’t think people should stop using cancer drugs that are saving lives,” said Addison, co-director of the Cardio-Oncology Program at the Ohio State University Comprehensive Cancer Center–James Cancer Hospital and Solove Research Institute, Columbus, Ohio. Rather, clinicians treating cancer patients should use a lower threshold for initiating a cardiovascular workup “if a patient is showing signs or symptoms of a potential heart issue.”
According to the researchers, cardiovascular events have become increasingly common with the use of cancer therapies, but “the prevalence and factors associated with these warnings are unknown.”
The authors found that of 125 FDA-approved cancer treatments, cardiovascular disease warning were issued for 32 (25.6%). Multiple warnings were issued for two dozen therapies (21.6%). Warnings were issued for more than 40% of the immune and targeted therapies — a total of 15 drugs.
- After arrhythmias (23.5%),
- uncontrolled hypertension (12%) and
- heart failure (11.4%)
were the most common reasons for a cardiac safety communication…
Addison noted that the average timing of cardiac safety communications masks wide variations by event type. Alerts regarding heart failure or hypertension, for example, emerged around 1200 days after a drug approval, but it took more than 2000 days for alerts about arrhythmias to be issued.
The authors said that the “delayed recognition is concerning, particularly given the rapid emergence of many targeted and immune-based cancer therapies, and the potentially devastating consequences of cardiotoxic events…”
“In a lot of cases, these newer drugs are really changing how we treat cancer patients,” said Eiman Jahangir, MD, MPH. “And typically, the benefit of curing a cancer outweighs the risk of a potential cardiac outcome, but as clinicians, we also need to know what [risks] to look for. If we know [about the risk for cardiac events], then we’re more likely to pick them up when a patient presents with symptoms.”
Footnotes-
- Chemotherapy-induced cardiomyopathy-https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3318862/
- Chemotherapy-induced atrial fibrillation – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6198283/
- Chemotherapy-induced hypertension- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3113122/
- Valvular Damage https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7293871/
- Chemotherapy-induced Thrombosis- https://pubmed.ncbi.nlm.nih.gov/16388837/