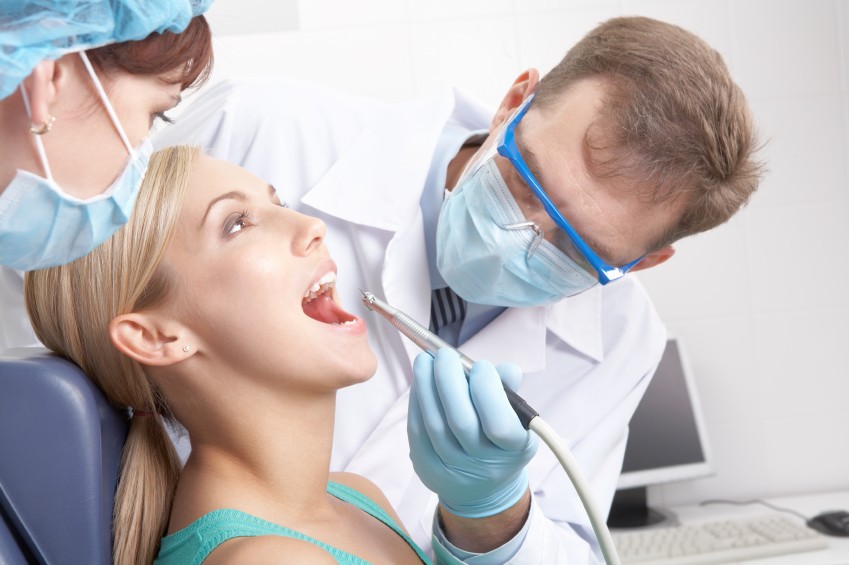
Patients who have received radiation for head/neck cancer often suffer from the unpleasant and distressing side-effect of a dry mouth, caused by damage to their salivary glands from the radiation
Xerostomia, also known as dry mouth, is the most serious side effect of radiation for head/neck cancer that you have never heard of. If a person has a serious enough case of this side effect, their gums will recede to a point where their teeth can fall out. You don’t want this to happen to you…
I don’t have head/neck cancer. I have multiple myeloma. My cancer presented itself by a lesion growing in my fifth cervical vertebra. The lesion was removed, my neck was surgically stabilized and I had local radiation to the area.
That was in the spring of ’94. In the years following this procedure I worked to manage my myeloma, manage side effects such as cataracts, chemobrain, nerve damage, irritable bladder and manage my risk of relapse or secondary cancers but I never gave a passing thought to dry mouth.
Skip ahead to today- 6/27/13. My gums have receded and the enamel on my teeth is slowly eroding. Dry mouth is sneaky. I wish I knew then what I know now. I read the study below about acupuncture healing dry mouth about five years after I had begun struggling with dry mouth. I found an acupuncturist in my town and underwent this therapy for about six months. Long-story short, my dry mouth is no-more.
My advice is that if you have undergone radiation therapy anywhere near your mouth and you begin waking up at night or in the morning with a really, really dry mouth then
- buy a dry mouth mouthwash such as Bioperene for temporary relief and
- keep a spray bottle of Bioperene on your bedside table for quick late-night therapy
and find an acupuncturist in your area to see regularly.
I am both a cancer survivor and cancer coach. To learn more about dry mouth and other possible side effects and how to prevent them, scroll down the page, post a comment or question and I will reply ASAP.
Thanks and good luck.
David Emerson
- Cancer Survivor
- Cancer Coach
- Director PeopleBeatingCancer
Recommended Reading:
“Xerostomia, also known as dry mouth[1] and dry mouth syndrome,[2] is dryness in the mouth, which may be associated with a change in the composition of saliva, or reduced salivary flow, or have no identifiable cause…
“Patients who have received radiotherapy for head and neck cancer often suffer from the unpleasant and distressing side-effect of a dry mouth, caused by damage to their salivary glands from the radiation…Now, a new study has shown that acupuncture can relieve the symptoms of dry mouth (known as xerostomia).
Around half a million people worldwide develop head and neck cancer each year and, at present, there are few effective treatments for dry mouth, which is a common side effect of radiotherapy; as many as 41% of patients can still be suffering from it five years later. Xerostomia affects the patients’ quality of life, interfering with taste, chewing, speaking and sleeping. Short-term solutions such as mouthwashes, gels and toothpastes provide some respite, while treatment with a drug called pilocarpine has its own unwanted side-effects…”
The Most BioAvailable Curcumin Formulas
“Based on a review of these studies, it is evident that better bioavailability of formulated curcumin (CU) products is mostly attributed to improved solubility, stability, and possibly low first-pass metabolism”
A search of the Pubmed database for the word curcumin yields 601 studies spaning health topics from multiple myeloma and colorectal cancer, to chemotherapies that synergizes with CU, to Alzheimer’s Disease, arthritis and more. Based on years of reading studies and personal accounts, I think it is safe to say that CU supplementation is safe and relatively inexpensive.

I have read about myeloma patients taking daily doses of CU from 400 milligrams to 8 grams (1000 milligrams = 1 gram). By almost any measure, CU is a safe, inexpensive wonder drug.
The only challenge is that CU is famously difficult to absorb in the body. In other words, a person has to mix curcumin with some sort of fat (coconut oil, chocolate, etc.) or take a brand of curcumin capsule that is already formulated to be more “bioavailable” in order to derive the full benefit of CU.
The study linked and exerpted below reviews different formulations of CU. The study itself lists the three most bioavailable formulation/brand of CU and I’ve added an excerpt from a further review from Consumerlab.com that lists four additional bioavailable brands of CU.
Recommended Reading:
“CU is a bright yellow chemical produced by some plants. It is the principal curcuminoid of turmeric (Curcuma longa), a member of the ginger family, Zingiberaceae. It is sold as an herbal supplement, cosmetics ingredient, food flavoring, and food coloring.[1]“
“Curcumin is a widely studied natural compound which has shown tremendous in vitro therapeutic potential. Despite that, the clinical efficacy of the native CU is weak due to its low bioavailability and high metabolism in the gastrointestinal tract. During the last decade, researchers have come up with different formulations with a focus on improving the bioavailability of curcumin. As a result, a significant number of bioavailable curcumin-based formulations were introduced with the varying range of enhanced bioavailability.
The purpose of this review is to collate the published clinical studies of CU products with improved bioavailability over conventional (unformulated) CU. Based on the literature search, 11 curcumin formulations with available human bioavailability and pharmacokinetics data were included in this review. Further, the data on clinical study design, analytical method, pharmacokinetic parameters and other relevant details of each formulation were extracted.
Based on a review of these studies, it is evident that better bioavailability of formulated curcumin products is mostly attributed to improved solubility, stability, and possibly low first-pass metabolism. The review hopes to provide a quick reference guide for anyone looking information on these bioavailable curcumin formulations.
Based on the published reports,
exhibited over 100-fold higher bioavailability relative to reference unformulated CU. Suggested mechanisms accounting for improved bioavailability of the formulations and details on the bioanalysis methods are also discussed.”
According to Consumerlab.com:
“Novasol has the highest bioavailability (185 x compared to unforumulated CU), followed by Curcuwin (136 x), Longvida (100 x), Meriva (48 x), BCM-95 (27 x), Curcumin C3 Complex + Bioperene (20 x), and then Theracumin (16 x).”
APPIP ERROR: amazonproducts[
AccessDeniedAwsUsers|The Access Key Id AKIAJAJ37JVNL7OUU4CA is not enabled for accessing this version of Product Advertising API. Please migrate your credentials as referred here https://webservices.amazon.com/paapi5/documentation/migrating-your-product-advertising-api-account-from-your-aws-account.html.
]