Diagnosed with Cancer? Your two greatest challenges are understanding cancer and understanding possible side effects from chemo and radiation. Knowledge is Power!
Learn about conventional, complementary, and integrative therapies.
Dealing with treatment side effects? Learn about evidence-based therapies to alleviate your symptoms.
Click the orange button to the right to learn more.
- You are here:
- Home »
- Blog »
- Healthy Living Products »
- Bladder Cancer- Increase Overall Survival
Bladder Cancer- Increase Overall Survival
“61% of patients with Non-Muscle Invasive Bladder Cancer (NMIBC) will experience recurrence within the first year after initial resection, and up to 78% will experience recurrence within five years”
If you have been diagnosed with Non-Muscle Invasive Bladder Cancer then, according to the first study linked and excerpted below, chances are pretty good that you will relapse in the next 1-5 years (61% and 78% respectively).
The three studies linked and excerpted at the bottom of the page cite the ability of curcumin, resveratrol and omega 3 fatty acids to reduce your risk of relapse.
I am blogging about evidence-based, non-toxic therapies to reduce the risk of bladder cancer relapse because I have supplemented with these non-toxic therapies myself for years now. And I have remained in complete remission from my own “incurable” cancer, multiple myeloma, since 1999.
I am both a cancer survivor and cancer coach. I have learned that cancer patients and survivors must draw one the best of both conventional (FDA approved) and evidence-based non-conventional therapies in order to manage their cancers for the long-term.
I’m not talking about curing any type of cancer. I encourage all cancer survivors to eat well, exercise, supplement, detox, etc. In short, I encourage cancer survivors to focus on their health for the rest of their lives.
I take Life Extension Super Bio-Curcumin, Optimized Resveratrol and Super Omega 3 Fatty Acids for several reasons. They have all been evaluated and approved by Consumerlab.com, an independent testing service, they are all formulas that provide efficacy and value and I can order each easily and cost-effectively through Amazon Prime.
Please scroll down the page, post a question or comment and I will reply to you ASAP.
Thank you,
David Emerson
- Cancer Survivor
- Cancer Coach
- Director PeopleBeatingCancer
Recommended Reading:
- Cancer Coaching-Ballooning Survivor population in the U.S
- Bladder Cancer – Conventional with Non-Conventional
- Bladder Cancer- “No Sex Drive/no Desire for Sex at All…”
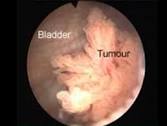
Enhanced Cystoscopy Improves the Care of Bladder Cancer Patients
“While survival rates for the majority of patients with non-muscle invasive bladder cancer (NMIBC) are favorable, bladder cancer has a relatively high risk of disease recurrence and progression that can significantly impact a patient’s quality of life and eventual cancer outcome.
Depending on the grade and stage at initial diagnosis, up to 61% of patients with NMIBC will experience recurrence within the first year after initial resection, and up to 78% will experience recurrence within five years.
In addition, there is a high risk for progression to muscle-invasive bladder cancer, with approximately 17% of patients progressing at one year and 45% progressing at five years.
Thus, the journey of patients with NMIBC is frequently characterized by multiple tumor recurrences and repeated therapeutic interventions over the course of their care, typically requiring lifelong monitoring. These statistics help to explain why BC is one of the most expensive cancers to manage, accounting for almost $3.7 billion in direct costs in the US each year.
Curcumin modulates micrRNA-203 mediated regulation of the Src-Akt axis in bladder cancer
“BC is often associated with recurrence and progression to invasive metastatic disease that have palliative therapeutic options. The use of traditional chemotherapeutic agents for BC management often suffer from toxicity and resistance concerns. This emphasizes the need for development of safer, natural, non-toxic compounds as chemotherapeutic/chemopreventive agents…
In conclusion, our study suggests that curcumin has the potential to be developed as an epigenetic agent for BC as it upregulates tumor suppressor miR-203 in bladder cancer by promoting DNA hypomethylation of the miR-203 promoter that in turn, leads to downregulation of its target oncogenes…”
Resveratrol induces apoptosis of bladder cancer cells via miR‑21 regulation of the Akt/Bcl‑2 signaling pathway.
“Resveratrol, an edible polyphenolic phytoalexin present in grapes and red wine, has been reported to inhibit proliferation and induce apoptosis of BC cells…
Resveratrol induced the cytotoxicity and apoptosis of T24 and 5637 cells in a dose‑dependent manner...Collectively, data revealed that the effect of resveratrol on BC cell apoptosis was due to miR‑21 regulation of the Akt/Bcl‑2 signaling pathway…”
Chemopreventive effect of omega-3 polyunsaturated fatty acids and atorvastatin in rats with bladder cancer.
“Bladder cancer remains a huge concern for the medical community because of its incidence and prevalence rates, as well as high percentage of recurrence and progression…
Collectively, omega-3 polyunsaturated fatty acids and atorvastatin, besides having anti-inflammatory properties, proved a chemopreventive effect against BC, which nominates them to be used as adjuvant therapy with other chemotherapeutics…”