
Diagnosed with Cancer? Your two greatest challenges are understanding cancer and understanding possible side effects from chemo and radiation. Knowledge is Power!
Learn about conventional, complementary, and integrative therapies.
Dealing with treatment side effects? Learn about evidence-based therapies to alleviate your symptoms.
Click the orange button to the right to learn more.
- You are here:
- Home »
- Blog »
- side effects ID and prevention »
- Cancer-Therapy-induced Cardiomyopathy and Your Genes
Cancer-Therapy-induced Cardiomyopathy and Your Genes
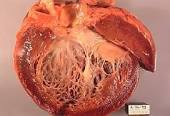
“The increased burden of rare variants including TTNtv, indicate that genetics are an important component in Cancer-Therapy-induced Cardiomyopathy (CCM) susceptibility and adverse outcomes”
I call it chemotherapy-induced cardiomyopathy or CIC. The first study linked below calls it cancer therapy-induced cardiomyopathy or CCM. I think I prefer CIC…
What everyone agrees on is the fact that many chemotherapy regimens are cardio toxic. Meaning these chemotherapies can cause heart damage. In my case, I flipped into CIC and chronic atrial fibrillation more than 15 years AFTER I underwent several different cardio toxic chemotherapy regimens.
My issue with both studies below are two-
First, that the patient’s genetic make-up pre-disposes him/her to CIC/CCM. While our genes may predispose a cancer patient to heart damage caused by chemo, let’s be clear. It is the chemo that is causing the heart damage.
Secondly, the possible solution proposed by doctors quoted in the articles simply talk about increased monitoring. Once I developed CIC and afib, monitoring me, was moot. The damage was done. My heart was NOT going back.
In my experience, I think that identifying those genes that predispose a patient to CIC should signal a number of evidence-based but non-toxic heart health therapies to come to the rescue.
Heart health therapies such as:
- exercise
- heart healthy nutrition
- heart healthy supplementation
- lifestyle therapies
I believe in evidence-based non-conventional therapies because I myself, have managed my CIC and Afib since 12/10 with these therapies in the absence of any conventional heart meds. None. Nada. Zip meds.
Did you undergo cardio toxic chemo? Do you have chemotherapy-induced cardiomyopathy? Scroll down the page, post a question or comment and I will reply to you ASAP.
To Learn More about DNA damage caused by chemotherapy and radiation- click now
Thanks,
David Emerson
- Cancer Survivor
- Cancer Coach
- Director PeopleBeatingCancer
- Coenzyme Q10 Is Safe and Effective Cardiomyopathy Therapy
- Chemotherapy-induced Cardiomyopathy- How long have I got?
- Taurine for Chemo-Induced Congestive Heart Failure (CHF)
Genetic Crosstalk in Cancer-Therapy-Induced Cardiomyopathy
“Patients with cancer-therapy-induced cardiomyopathy (CCM) have an excess of rare mutations in dilated cardiomyopathy genes, and towering among them is TTN, the gene encoding the massive sarcomere protein titin, new research suggests.
The study showed that 7.5% of 213 adult and pediatric CCM patients had truncating variants in TTN, compared with 1.1% of 2053 unselected breast or lung participants in the Cancer Genome Atlas (P = 7.36e-08) and 0.7% of 445 healthy volunteers in the UK Digital Heart Project (P = 3.42e-06).
Adults with CCM and TTN truncating variants (TTNtv) also had more heart failure hospitalizations and atrial fibrillation (P = .003) and worse cardiac function (P = .03) than those without the variants.
In addition to TTNtv, the analysis identified rare protein-altering variants in five dilated cardiomyopathy (DCM) genes: BAG3, LMNA, MYH7, TNNT2, and TCAP.
“The increased burden of rare variants including TTNtv, indicate that genetics are an important component in CCM susceptibility and adverse outcomes…”
“These data establish a genetic relationship between DCM and CCM,” they note. Cardiomyopathy variants were found in 12.2% of patients with CCM and occur in about 40% of patients with familial and sporadic DCM.
“The one thing that we are very excited about with this study is the potential implication of hopefully changing management of patients undergoing cancer therapy,” Kim told theheart.org | Medscape Cardiology.
“Once we identify who is going to be at higher risk, we can more closely monitor them, have more frequent cardiac monitoring, or perhaps take cardioprotective medications to improve the cardiovascular outcomes,” she said. “We believe that improving these cardiovascular outcomes will also improve their oncologic outcomes because nowadays, unfortunately, if someone develops cardiotoxicity, their oncologic regimen gets interrupted or just stopped.”
At Risk for CCM
Anthracyclines, used in nearly all CCM cases, cause cardiotoxicity in up to 10% of patients with cumulative dosages of 250 mg/m². When combined with other therapies, such as trastuzumab, depressed left ventricular ejection fraction (LVEF) occurs in up to 34% of patients and severe, symptomatic heart failure in 2% to 4%, the authors note. Female sex, older age, and pre-existing cardiac risk factors also contribute to CCM but “even when accounting for these factors, predicting individual susceptibility to CCM remains challenging.”
TTN truncating variants are prominent in DCM, occurring in 15% of ambulatory and 25% of end-stage patients but are rarely identified in childhood-onset DCM, according to the researchers. Here, 8.1% of adults and 5.0% of children with CCM harbored these deleterious variants…
Three-fourths of CCM patients were female, 90% received anthracyclines, and 33% of adults received trastuzumab. The median time to CCM diagnosis after starting cancer treatment was 3.0 years (range, 1–9 years) in the European cohort and 0.3 to 0.7 years in the US cohorts.
Across all cohorts, treatment with a high cumulative dose anthracycline (>400 mg/m²) was not associated with poorer LV function at CCM diagnosis (mean LVEF, 42%), the authors say.
Cardiac recovery was reported in about half of CCM patients from each cohort, but 9% of the European cohort underwent cardiac transplant. Cardiac death occurred in 3% of the European cohort and 5% of the US pediatric cohort…
“One thing we’d like to pursue in the near future is to look at other genes because we believe there will be other genetic contributors to cancer therapy-induced cardiomyopathy,” Kim said. “We believe that genes important in drug metabolism will be one of them” and “there are other additional genes that are involved in the signaling pathway that are also very important in cardiomyocytes function.”
Circulation. Published online April 16, 2019. Full text
Genetic variant linked to increased risk for heart disease in cancer survivors
“Patients who received the chemotherapy drug doxorubicin for childhood cancer and harbor a specific genetic mutation near the KCNK17 gene experienced significantly greater risk for heart disease later in life, study results have shown…
A retrospective analysis by investigators at St. Jude Children’s Research Hospital suggested cancer survivors who carry a single copy of the genetic variant rs2815063-A and received treatment with doxorubicin have a three-times higher risk for developing treatment-related cardiac dysfunction later in life as those who do not carry the allele…
Doxorubicin, an anthracycline-based chemotherapy, is part of the standard of care for many pediatric cancers, according to Yadav Sapkota, PhD, assistant member at St. Jude Children’s Research Hospital. The therapy is also known to cause cardiac dysfunction in long-term cancer survivors, he added…
“Many childhood cancer survivors develop cardiotoxicity, but there is a variation in risk, suggesting a potential role of genetic susceptibility,”


