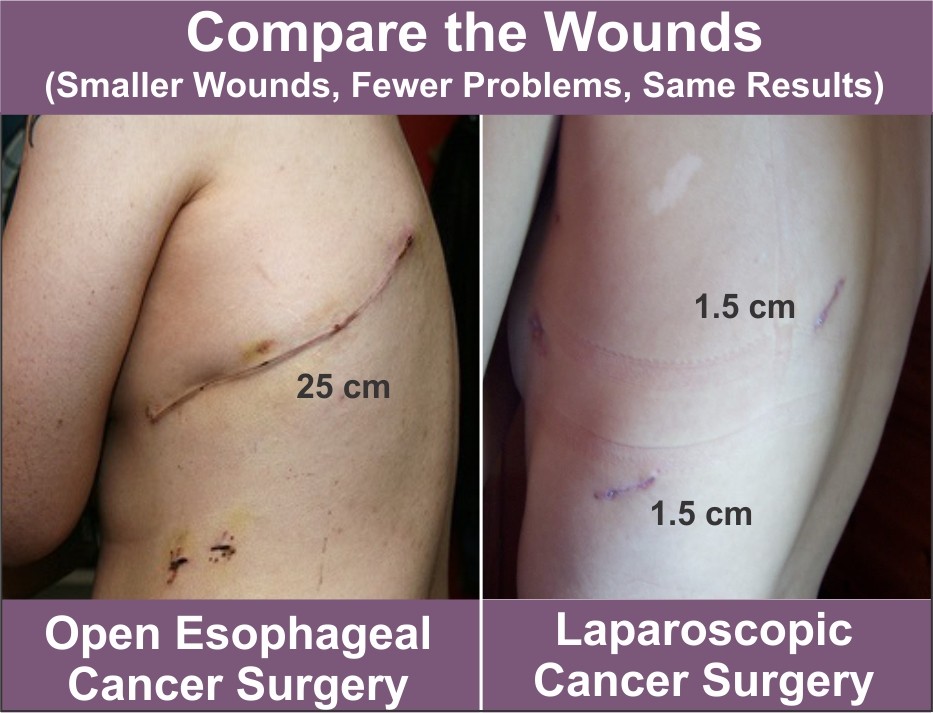
Pre-habilitation or to To Pre-habilitate is “getting in shape” for your surgery is once such low-tech but beneficial therapy. Where newly diagnosed cancer is concerned, surgery is often the best, first step in debulking a host of cancers including including cancers of the
- esophagus,
- stomach,
- biliary system,
- pancreas,
- small intestine,
- large intestine,
- rectum
- and anus.
Hospitals today love to advertise how high-tech they are. With newly diagnosed cancer patients however, combine both high-tech therapies with evidence-based low-tech therapies.
Understand that some of these cancers have a discouraging 5 year survival rate. Meaning you need to use the most effective evidence-based therapies possible- high or low tech.
Debulking cancer by surgically removing as many cancer cells as possible can be extremely effective therapy for the cancer patient.
The article linked and excerpted below reports patients that supplement with Omega-3 polyunsaturated fatty acids (n-3 PUFAs), on average, do better than surgical patients who do not.
Keep in mind that conventional oncology focuses on the “standard-of-care” or those therapies approved by the FDA. Which is not to say that both high-tech and low-tech therapies are important to the average cancer patient.
Have you been diagnosed with a GI Tract Cancer? Are you considering surgery? To learn more about therapies that can improve both your quality and length of life scroll down the page, post a question or comment and I will reply to you ASAP.
To Learn More about Esophageal Cancer- click now
Thanks
David Emerson
- Cancer Survivor
- Cancer Coach
- Director PeopleBeatingCancer
Recommended Reading:
Introduction: Prehabilitation prior to major surgery has increased in popularity over recent years and aims to improve pre-operative conditioning of patients to improve post-operative outcomes. The beneficial effect of such protocols is not well established with conflicting results reported. This review aimed to assess the effect of prehabilitation on post-operative outcome after major abdominal surgery.
Methods: EMBASE, Medline, PubMed and the Cochrane database were searched in August 2018 for trials comparing outcomes of patients undergoing prehabilitation involving prescribed respiratory and exercise interventions prior to abdominal surgery. Study characteristics, overall and pulmonary morbidity, length of stay (LOS), maximum inspiratory pressure and change in six-minute walking test (6MWT) distance were obtained. The primary outcome was post-operative overall morbidity within 30 days. Dichotomous data were analysed by fixed or random effects odds ratio. Continuous data were analysed with weighted mean difference.
Results: Fifteen RCTs were included in the analysis with 457 prehabilitation patients and 450 control group patients. A significant reduction in overall (OR 0.63 95% CI 0.46-0.87 I2 34%, p = 0.005) and pulmonary morbidity (OR 0.4 95% CI 0.23-0.68, I2 = 0%, p = 0.0007) was observed in the prehabilitation group. No significant difference in LOS (WMD -2.39 95% CI -4.86 to 0.08 I2 = 0%, p = 0.06) or change in 6MWT distance (WMD 9.06 95% CI -35.68, 53.81 I2 = 88%, p = 0.69) was observed.
Conclusions: Prehabilitation can reduce overall and pulmonary morbidity following surgery and could be utilised routinely. The precise protocol of prehabilitation has not been completely established. Further work is required to tailor optimal prehabilitation protocols for specific operative procedures.
“Background- Surgical resection remains the primary treatment for gastrointestinal (GI) malignancy including early-stage cancer. Omega-3 polyunsaturated fatty acids (n-3 PUFAs) have been reported to have beneficial clinical and immune-modulating effects in the prognosis of GI cancer patients undergoing surgery.
Methods- We searched PubMed, Embase, EBSCO-Medline, Cochrane Central Register of Controlled Trials (CENTRAL), CNKI and Wanfang to identify primary research reporting the effects of n-3 PUFAs compared with isocaloric nutrition on GI cancer patients who underwent surgery up to the end of June 30, 2016. Two authors independently reviewed and selected eligible randomized controlled trials (RCTs).
Results-A total of 9 RCTs (623 participants) were included. The n-3 PUFAs regime resulted in lower levels of C-reactive protein (CRP) (P < 0.05), interleukin-6 (IL-6) (P < 0.01), and higher levels of albumin (ALB), CD3+ T cells, CD4+ T cells and CD4+/CD8+ ratio (P < 0.05) compared with the isocaloric nutrition regime. However, there was no significant difference in the level of tumor necrosis factor-α (TNF-α) between the n-3 PUFAs regime and the isocaloric nutrition regime (P = 0.17). And the level of CD8 + T cells decreased compared with the isocaloric nutrition regime (P < 0.0001).
Conclusions-Our meta-analysis revealed that n-3 PUFAs are effective in improving the nutritional status and immune function of GI cancer patients undergoing surgery as they effectively enhance immunity and attenuate the inflammatory response.
“Gastrointestinal cancer refers to malignant conditions of the gastrointestinal tract (GI tract) and accessory organs of digestion, including the esophagus, stomach, biliary system, pancreas, small intestine, large intestine, rectum and anus. The symptoms relate to the organ affected and can include obstruction (leading to difficulty swallowing or defecating), abnormal bleeding or other associated problems…”