The studies linked and excerpted below cite intravenous vitamin C therapy for multiple myeloma as being safe and effective-
David- You have done a lot of high dose intravenous Vitamin C (IVC) infusions as a multiple myeloma therapy, what are your thoughts on the risk of kidney stones or some damage to your liver if you do IVC, High dose injections?
Integrative doctors and Haemotologists/Oncologists warn against it.
Your final thoughts please?
Thank you. Mac
Dear Mac-
I have never undergone intravenous vitamin C therapy (IVC) for my multiple myeloma. I stir a vitamin C powder (2500 mg) w/ amino acids into my juice each am. The powder has been shown to be a therapy for my chemo-induced cardiomyopathy. I think oral vitamin C therapy is generally a positive for our immune function.
I have blogged about IVC therapy for early stage multiple myeloma patients. I have written about IVC as a therapy once a multiple myeloma patient has achieved remission in order to deepen and lengthen his/her remission.
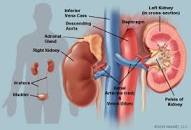
I have read about the idea of, the possibility of kidney involvement with IVC. Whether we are talking about kidney stones or serum creatinine or eGFR, my overall concern for MM patients and survivors would be IVC interfering with kidney function in some way.
If you notice, the study excerpted below studies not just “kidney stones” yes or no but also kidney function as evidenced by serum creatinine and eGFR. MM patients and survivors get blood and urine testing done routinely. Monitoring kidney function is an important aspect of MM as freelight chains, chemo, bisphosphonate therapy, contrast agents, others, all have been shown to negatively affect kidney function.
Based on what I have read about IVC and kidney stones or kidney damage, I don’t see IVC as being a problem. Yes, all MM patients and survivors should undergo blood and urine testing regularly. But I don’t see IVC as being a problem for kidney function.
David Emerson
- MM Survivor
- MM Cancer Coach
- Director PeopleBeatingCancer
“We carried out a prospective case series study of 157 adult patients who commenced IVC therapy at Integrated Health Options clinic between 1 September 2011 and 31 August 2012, with follow-up for 12 months.
Inquiries into the occurrence of renal stones were conducted at enrolment, 6 and 12 months, and renal function blood tests were conducted at enrolment, 4 weeks and every 12 weeks thereafter in a subgroup of patients.
No renal stones were reported by any patients in the study, despite 8% of the patients having a history of renal stones. In addition, the majority of patients investigated had stable renal function during the study period as evidenced by little change in serum creatinine levels and estimated glomerular filtration rate (eGFR) following IVC.
In conclusion, IVC therapy was not associated with patient-reported renal stones. Although not the primary focus of this study, it was also observed that there was no significant change in mean serum creatinine or eGFR for those who had follow-up renal function blood tests…
Follow-Up
Renal function blood tests were routinely ordered for all patients prior to commencing IVC therapy and repeated after 4 weeks then every 12 weeks for as long as the patient continued to have IVC treatments at the clinic. Changes in renal function were managed in such a way that if the eGFR reduced 15–20%, renal function blood tests were repeated within 1–2 weeks, and if the eGFR reduced more than 20%, IVC was promptly ceased, the patient reviewed and any possible causes addressed [
28,
29]. Renal function tests were repeated 1–2 weeks later and if normalized or improving, IVC was restarted and monitored closely until stabilized. If levels continued to reduce, IVC was not restarted…
Enrolled patients were contacted by phone 6 months and 12 months after admission and asked a standardised question about experiencing, or seeking medical advice for, symptoms of renal stones or renal colic since starting IVC. If a patient died during the follow-up period, a coroner’s report or death certificate was requested to check whether renal stones or renal impairment/failure was the cause of death or the antecedent cause of death. Due to a significant proportion of patients presenting with advanced cancer, a number of patients died during the study period (37/157, or 24%). Some patients were lost to follow-up due to moving overseas or lost to contact (three at six months, and six at 12 months)…
Outcome Data
Six months after commencing IVC therapy, 153 out of 157 patients (124 alive patients and 29 deceased patients) had no reported renal stones or no renal stones causing renal impairment/failure recorded on death certificates ().
Discussion
The primary outcome of this prospective study was that no renal stones were reported by any patient who received IVC during the study period, despite 8% of participants reporting previous episodes of renal stones. This study showed that IVC was not associated with any cases of symptomatic renal stones or renal stones causing renal failure/death in 100% of patients able to be followed up over a 12-month period (94% follow-up).
Although not the focus of this study, half of the eligible patients had follow-up creatinine and eGFR results available. Of the patients that provided both before and after renal function blood tests (50% of the patients), IVC therapy was not associated with a significant detrimental effect on renal function.
“Finally, retrospective cohort study showed that intravenous VC use was linked to improved disease-free survival (DFS) in HCC patients. Our data highlight that pharmacologic VC can effectively kill liver cancer cells and preferentially eradicate liver CSCs, which provide further evidence supporting VC as a novel therapeutic strategy for HCC treatment…”