
Diagnosed with SMM, SPB, or MGUS?
Learn how you can stall the development of full-blown Multiple Myeloma with evidence-based nutritional and supplementation therapies.
Click the orange button to the right to learn more.
- You are here:
- Home »
- Blog »
- Pre-Myeloma »
- MGUS, PPI’s, Barrett’s Esophagus and Bone Health
MGUS, PPI’s, Barrett’s Esophagus and Bone Health
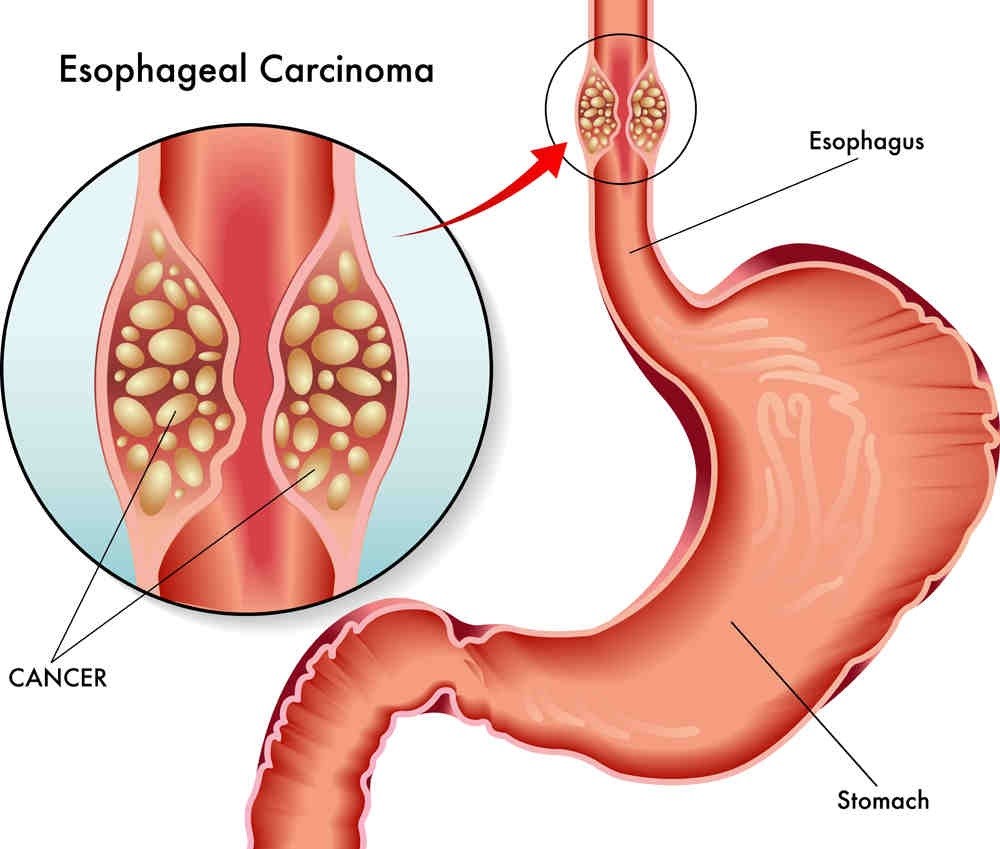
Esophageal involvement (barrett’s esophagus) by multiple myelomas is uncommon in the English literature. Here we report an extremely rare involvement in MM/MGUS
Hi David- I was diagnosed with Barrett’s Esophagus in 2000 and was on Proton Pump Inhibitors for over 15 years. I finally got off them 18 months ago and switched to Zantac (three 75mgs tabs a day).
 I had an initial return of symptoms, but these calmed down fairly quickly with the Zantac. I think the tendency to have a rebound effect is a very scary issue. I had tried to stop several times before and restarted due to pain.
I had an initial return of symptoms, but these calmed down fairly quickly with the Zantac. I think the tendency to have a rebound effect is a very scary issue. I had tried to stop several times before and restarted due to pain.
But the truth is once I got through that stage I have been really comfy on my new regime.
I have had two episodes of pneumonia in tbe past and issues with weight gain, poor circulation in my legs and muscle loss/pain in my arms. I have also had memory problems. Most of these issues have begun to improve since stopping the PPI. I also have MGUS so am at risk for Multiple Myeloma and wonder if MGUS/MM could be another risk with taking PPIs? Richard
Hi Richard- I am sorry to learn of your various health challenges.
Several things. Long-term PPI use is associated with several serious health problems. I would have to do more research but I believe that it is reasonable to believe that at least some of your health challenges are related to your long-term PPI use.
There may be a link between your MGUS and BE-
If you have discontinued PPI’s I think the real question is the relation between Zantac and MGUS and MM. As you can read from the study linked below Zantac use can prevent abosorbtion of minerals. This reduced mineral absorbtion can weaken your bones. MGUS can weaken bones.
Bone mineral density in patients taking H2-receptor antagonist.
In short, long-term PPI use causes a host of health issues including malabsorbtion of minerals. MGUS is a blood disorder but it can lead to bone health problems. I cannot say if PPI use leads to MGUS…
I encourage you to consider the Pre-Myeloma Cancer Coaching program.
To learn more about the evidence-based protocols you can follow to prevent your Pre-Myeloma from becoming Multiple Myeloma, please watch the short video below:
Click here to get the FREE Pre-Myeloma Introduction Guide and follow along.
Click here to get the FREE Pre-Myeloma First Questions Guide.
Not only will you undergo bone health therapies but one key supplement called curcumin reduces the risk of MM as well as reduces the risk of your BE becoming esophageal cancer.
Let me know if you have any questions.
Thanks and hang in there,
David Emerson
- Cancer Survivor
- Cancer Coach
- Director PeopleBeatingCancer
Recommended Reading:
- Cancer Coaching- Nutrition = anti-inflammation, angiogenesis, antioxidation
- Curcumin Lowers Blood Pressure/Hypertension
The Most BioAvailable Curcumin Formulas
“Based on a review of these studies, it is evident that better bioavailability of formulated curcumin (CU) products is mostly attributed to improved solubility, stability, and possibly low first-pass metabolism”
A search of the Pubmed database for the word curcumin yields 601 studies spaning health topics from multiple myeloma and colorectal cancer, to chemotherapies that synergizes with CU, to Alzheimer’s Disease, arthritis and more. Based on years of reading studies and personal accounts, I think it is safe to say that CU supplementation is safe and relatively inexpensive.
I have read about myeloma patients taking daily doses of CU from 400 milligrams to 8 grams (1000 milligrams = 1 gram). By almost any measure, CU is a safe, inexpensive wonder drug.
The only challenge is that CU is famously difficult to absorb in the body. In other words, a person has to mix curcumin with some sort of fat (coconut oil, chocolate, etc.) or take a brand of curcumin capsule that is already formulated to be more “bioavailable” in order to derive the full benefit of CU.
The study linked and exerpted below reviews different formulations of CU. The study itself lists the three most bioavailable formulation/brand of CU and I’ve added an excerpt from a further review from Consumerlab.com that lists four additional bioavailable brands of CU.
Recommended Reading:
- CU versus Turmeric as Cancer Therapy-What’s the Difference?
- CU Suppresses Colon Cancer, Enhances Chemotherapy
- Prostate Cancer: CU Curbs Prostate Cancer Metastases, Study Shows
- CU vs. cancer: The scientific evidence continues to pile up
Curcumin
“CU is a bright yellow chemical produced by some plants. It is the principal curcuminoid of turmeric (Curcuma longa), a member of the ginger family, Zingiberaceae. It is sold as an herbal supplement, cosmetics ingredient, food flavoring, and food coloring.[1]“
Bioavailable curcumin formulations: A review of pharmacokinetic studies in healthy volunteers.
“Curcumin is a widely studied natural compound which has shown tremendous in vitro therapeutic potential. Despite that, the clinical efficacy of the native CU is weak due to its low bioavailability and high metabolism in the gastrointestinal tract. During the last decade, researchers have come up with different formulations with a focus on improving the bioavailability of curcumin. As a result, a significant number of bioavailable curcumin-based formulations were introduced with the varying range of enhanced bioavailability.
The purpose of this review is to collate the published clinical studies of CU products with improved bioavailability over conventional (unformulated) CU. Based on the literature search, 11 curcumin formulations with available human bioavailability and pharmacokinetics data were included in this review. Further, the data on clinical study design, analytical method, pharmacokinetic parameters and other relevant details of each formulation were extracted.
Based on a review of these studies, it is evident that better bioavailability of formulated curcumin products is mostly attributed to improved solubility, stability, and possibly low first-pass metabolism. The review hopes to provide a quick reference guide for anyone looking information on these bioavailable curcumin formulations.
Based on the published reports,
exhibited over 100-fold higher bioavailability relative to reference unformulated CU. Suggested mechanisms accounting for improved bioavailability of the formulations and details on the bioanalysis methods are also discussed.”
According to Consumerlab.com:
“Novasol has the highest bioavailability (185 x compared to unforumulated CU), followed by Curcuwin (136 x), Longvida (100 x), Meriva (48 x), BCM-95 (27 x), Curcumin C3 Complex + Bioperene (20 x), and then Theracumin (16 x).”



