Recently Diagnosed or Relapsed? Stop Looking For a Miracle Cure, and Use Evidence-Based Therapies To Enhance Your Treatment and Prolong Your Remission
Multiple Myeloma an incurable disease, but I have spent the last 25 years in remission using a blend of conventional oncology and evidence-based nutrition, supplementation, and lifestyle therapies from peer-reviewed studies that your oncologist probably hasn't told you about.
Click the orange button to the right to learn more about what you can start doing today.
- You are here:
- Home »
- Blog »
- Multiple Myeloma »
- Multiple Myeloma Chemotherapy- Kyprolis/Carfilzomib Heart Damage
Multiple Myeloma Chemotherapy- Kyprolis/Carfilzomib Heart Damage

Question-What is the rate of cardiovascular adverse events (CVAE) among patients receiving carfilzomib for multiple myeloma?
Hi David I told you a few months ago that I was diagnosed with myeloma on 23rd December 2009 and since then have had no treatment or symptoms. I have done lots of things to keep the treatment at bay including numerous supplements and various complementary and alternative therapies.
I have blood tests at hospital every two months. I’ve now been told that my haemoglobin is 89 and my light chains are around 1000.
The medics are now pressurising me even more into having treatment followed by a stem cell transplant.
I still feel determined to avoid treatment but don’t want to be stupid about it. Would appreciate your comments.
Recommended Reading:
- Healing Chemotherapy-Induced Heart Disease-
- Myeloma Survival- Low-dose Maintenance Therapy
- Multiple Myeloma Chemotherapy- Can Antioxidant Supplements Interfere w/ Chemo?
Carfilzomib-Associated Cardiovascular Adverse Events-
A Systematic Review and Meta-analysis
“QuestionWhat is the rate of cardiovascular adverse events (CVAE) among patients receiving carfilzomib for multiple myeloma?
FindingsIn this systematic review and meta-analysis of 24 prospective clinical trials including 2594 patients, all-grade and high-grade CVAE were seen in 18.1% and 8.2% of patients, respectively; cardiovascular adverse event rates were higher in trials using carfilzomib doses of 45 mg/m2 or more. In randomized clinical trials, the rate of high-grade CVAEs was more than double in carfilzomib treated patients than in controls.
MeaningCarfilzomib is associated with a high rate of clinically significant CVAEs, which underscores a need for increased awareness of these adverse events and further study into optimal patient selection and risk mitigation strategies
Clinical Evidence for Q10 Coenzyme Supplementation in Heart Failure: From Energetics to Functional Improvement
Multiple Myeloma Chemotherapy- Proteasome Inhibitors
“More importantly, a low concentration of RSV was synergistic with a low dose of the proteasome inhibitor carfilzomib (CFZ) to induce apoptosis in myeloma cells…
Bortezomib (velcade), Carfilzomib (kyprolis) and Ixazomib (ninlaro) are all proteasome inhibitors. All three chemotherapy drugs are multiple myeloma chemotherapy regimens approved by the FDA.
The challenge for multiple myeloma survivors however, is that each proteasome inhibitor will stop working eventually. MDR aka multi-drug resistance is a fact for all multiple myeloma chemotherapy regimens.
If you are a MM patient or survivor who has undergone several multiple myeloma chemotherapy regimens already and is wondering about your next therapy steps, consider integrative therapies- combinations of conventional and non-conventional proteasome inhibitors that integrate or enhance each other…”
“An analysis of past studies shows 18 percent of multiple myeloma patients receiving carfilzomib experience cardiovascular adverse events (CVAE) such as hypertension, heart failure, heart attacks, or arrhythmia…”
When I read the article below citing cardiovascular events in Carfilzomib/Kyprolis treatment, I found myself wondering how many times I had blogged about MMers, multiple myeloma side effects and chemotherapy-induced cardiotoxidity in the past. I mean, is it just me or do MMer’s sustain above average amounts of damage to their heart when they undergo chemotherapy? And can anything be done to minimize the damage?
Its important to understand that the cardiotoxicity of certain chemotherapy regimens is not necessarily zero-sum. Meaning, the multiple myeloma side effect discussed below is not like a light switch, on or off.
The cardiovascular adverse events (CVAE) discussed below occurs on a sectrum. Because the average age of newly diagnosed MMers is 67 according to the American Cancer Society, MMers as a group may be vulnerable to cardiovascular events aka heart damage.
A MMer can experience heart damage from Carfilzomib and other toxic therapies without knowing it immediately. That’s what happed to me. To quote the article below “Previous studies have shown nearly two-thirds of MM patients had cardiovascular disease at baseline, and 70 percent experienced cardiovascular events within six years...”
I underwent five courses of V.A.D. induction therapy in the spring of 1995, and two courses of cytoxan during he following September, and my doctor told me that my heart function was “within normal limits.”
As the doctor says in the article, “If you’re not specifically looking for this, you might report it differently,” Waxman said”
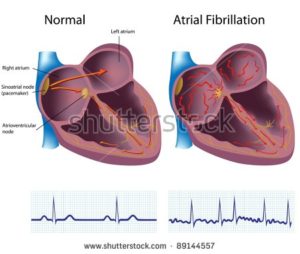
I developed chronic atrial fibrillation (AFib) in the fall of 2010, 15 years after the cardiologist told my oncologist that I was fine.
To be fair, I understand the need for toxic cancer killing therapies. MM can be an aggressive, difficult to control blood cancer. What bothers me is that 1) there are evidence-based therapies that reduce or prevent cardiovascular adverse events and 2) the oncologist should discuss this dramatic a side effect with the patient. The patient might want to reduce his or her risk of CVAE by lowering the dose of a cardiotoxic chemotherapy.
When I underwent adriamycin (the A in VAD), I wish that I knew that the supplement CoQ10 protected the heart from adriamycin cardiotoxicity.
- Curcumin and Doxorubicin Combo in Relapsed/Refractory Multiple Myeloma
- Multiple Myeloma-Atrial fibrillation, heart damage and chemoradiation
- Kyprolis vs. Bortizomib heart damage in Multiple Myeloma
After reading multiple studies about the cardiotoxicity of MM chemotherapy regimens, I came to the conclusion that if an MMer, any MMer, underwent multiple chemotherapy regimens over the course of their life as an MMer, they would suffer multiple assaults to their heart. The odds of identifying a problem with your heart increases with each course of chemotherapy, with each relapse. If you don’t experience heart damage during your induction therapy, you might have an episode during your ASCT. If not during your ASCT… well, you get the idea.
I developed chronic A-Fib after 1) two different induction therapies and 2) an ASCT and fifteen uneventful years.
Recommended Reading:
- Multiple Myeloma- The Cure vs. Control Debate
- A Different kind of Second Opinion-Multiple Myeloma Cancer Coaching
- CoQ10 (coenzyme Q 10) ubiquinone for chemo-induced heart damage
Carfilzomib Can Lead to Cardiovascular Toxicity in Multiple Myeloma Patients
“An analysis of past studies shows 18 percent of multiple myeloma patients receiving carfilzomib experience cardiovascular adverse events (CVAE) such as hypertension, heart failure, heart attacks, or arrhythmia. More than eight percent of patients experience high-grade CVAEs that are more severe, which is more than twice as common as with other drugs for treating relapsed myeloma…
Brendan M. Weiss, MD, an adjunct professor of Hematology Oncology at Penn, is the study’s senior author. Weiss also works in research and development at Janssen Pharmaceuticals, which does not manufacture or support any of the drugs involved in this analysis…
Researchers gathered data from 24 studies reported from 2007 through 2017, which included information on 2,594 MM patients. They found 18.1 percent of patients who took carfilzomib experienced CVAE, with 8.2 percent of those cases being grade three or higher, meaning they are categorized as severe. For comparison, a similar review of bortezomib, another proteasome inhibitor, found just 3.8 percent of patients experienced CVAE and only 2.3 percent were severe…
Researchers also found that higher doses of carfilzomib are associated with higher rates of CVAE, and that carfilzomib was associated with an elevated risk of CVAE compared to control groups who did not receive carfilzomib…
Researchers say these findings are particularly important since there are already overlapping risk factors for both MM and cardiovascular disease, such as older age and obesity. Previous studies have shown nearly two-thirds of MM patients had cardiovascular disease at baseline, and 70 percent experienced cardiovascular events within six years…
Researchers also called for further clinical trials to specifically evaluate this connection, arguing that it may be underrepresented by current data.
“If you’re not specifically looking for this, you might report it differently,” Waxman said.”
Cardiotoxicity Risk with Bortezomib Versus Lenalidomide for Treatment of Multiple Myeloma: A Propensity-Matched Study of 1,128 Patients
“Conclusion: The current study shows that bortezomib use in patients treated for multiple myeloma is associated with an increased risk of hospitalization for heart failure, but not acute MI or arrhythmia. Those with HF prior to initiating therapy with bortezomib had a significantly increased risk of hospitalization for HF when compared to those treated with lenalidomide. There was no significant difference in the rate of hospitalization for HF in those without a baseline diagnosis of HF. Collectively, these data have important implications for the management of patients with multiple myeloma, especially those with a history of HF at the time of therapy initiation…”