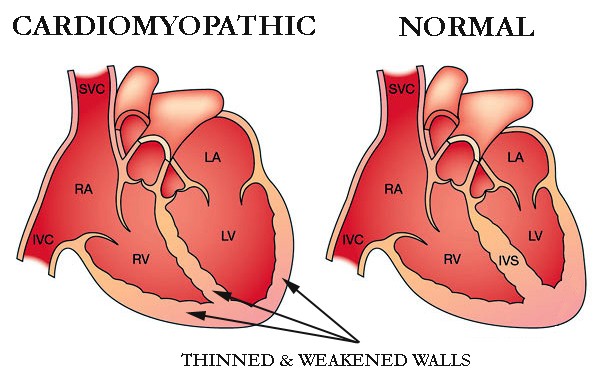
Multiple Myeloma-Anticipation of the period of greatest risk and recognition of patients with subclinical myocardial dysfunction may prevent clinical heart failure (HF)-
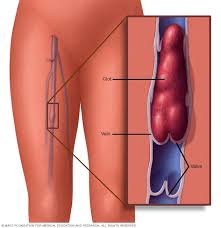
I underwent an autologous stem cell transplant for my multiple myeloma in December of 1995. I developed a DVT (blood clot) in 4/95 and again in the other leg several years later. I developed atrial fibrillation (A-fib) in late 2010. I was formally diagnosed with chemotherapy-induced cardiomyopathy (CIC)-heart failure (HF) in January of 2019.

My diagnosis of chemotherapy-induced heart failure in 2019 led to more focused research on the short, long-term and late stage adverse events that aggressive, high-dose chemotherapy can cause to the MM patient. As it happens, chemotherapy can be pretty damaging to your heart and cause damage to related systems-
DVT/blood clots,
a-fib- atrial fibrillation,
increased blood pressure,
weight gain, etc.
Though I have come across numerous studies that pre-date my treatment, documenting the short and long-term cardiotoxic effects of my induction, consolidation and ASCT chemotherapy regimens, my oncologist told me NOTHING about any of these probably adverse events.
The reason for oncology’s general attitude toward the heart failure associated with stem cell transplantation, at least as far as I can determine, is embodied in the phrase evidence-based medicine. To be more specific, what conventional oncology considers to be evidence-based is different from what I consider to be evidenced-based.
In the case of my deep-vein thrombosis aka DVT, had I been supplementing with omega-3 fatty acids, curcumin and enzymes, for example, I may not have developed my blood clot. In the case of my heart, had I been exercising, eating heart-healthy foods and taking with the above mentioned supplements, again, I would have dramatically reduced my risk of atrial fibrillation and heart failure.
Conventional oncology doesn’t not consider the numerous studies that document the benefits of nutrition, lifestyle and supplementation to be evidence-based. After researching my multiple myeloma and many short, long-term and late stage adverse events, I do consider these studies and associated therapies to be evidence-based medicine.
To be clear, I believe that MM patients need to understand both conventional or FDA approved therapies as well as evidence-based, non-conventional therapies.
I wish I knew then what I know now.
To learn more about both conventional and non-conventional evidence-based therapies for multiple myeloma and associated adverse events, scroll down the page, post a question or comment and I will reply to you ASAP.
Thank you,
David Emerson
- MM Survivor
- MM Cancer Coach
- Director PeopleBeatingCancer
Recommended Reading:
“Objective-To investigate subacute cardiac toxicity in patients with normal baseline cardiac function following autologous haematopoietic stem cell transplantation.
PATIENT AND METHODS:
Thirty-two consecutive patients (mean (SD) age 60 (11) years) with normal left ventricular ejection fraction (LVEF >or=50%) undergoing autologous haematopoietic stem cell transplantation were studied. Transthoracic echocardiography (including colour tissue Doppler imaging-derived myocardial velocities, strain and strain rates), troponin-T and B-type natriuretic peptide (BNP) and clinical details were recorded at baseline, after conditioning chemotherapy and serially over 6 weeks from the day of transplantation.
RESULTS:
The mean (SD) LVEF at baseline was 62 (6)% and decreased to 55 (16)%, 6 weeks after transplantation (p = 0.007). Cardiac toxicity (>or=10% absolute decline of LVEF to an LVEF <or=50%) developed in 10 (31%) patients within 17 (8) days of transplantation and was usually reversible. In these 10 patients, the nadir LVEF was 39 (12)%. Two patients developed severe clinical pulmonary oedema, one episode of which was fatal. Troponin-T was mildly raised in only three of the patients with cardiac toxicity. Peak BNP values were similar in patients with or without post-transplant cardiac toxicity (149 (100) vs 196 (178) pg/ml, p = 0.43). Multivariate Cox proportional hazard regression identified baseline mitral annular systolic velocity as the only independent predictor of cardiac toxicity (hazard ratio 0.42, 95% confidence interval 0.20 to 0.86, p = 0.02).
CONCLUSION:
Subacute cardiac toxicity is common after autologous haematopoietic stem cell transplantation, even in patients with apparently normal left ventricular function. Anticipation of the period of greatest risk and recognition of patients with subclinical myocardial dysfunction may prevent clinical heart failure.