Recently Diagnosed or Relapsed? Stop Looking For a Miracle Cure, and Use Evidence-Based Therapies To Enhance Your Treatment and Prolong Your Remission
Multiple Myeloma an incurable disease, but I have spent the last 25 years in remission using a blend of conventional oncology and evidence-based nutrition, supplementation, and lifestyle therapies from peer-reviewed studies that your oncologist probably hasn't told you about.
Click the orange button to the right to learn more about what you can start doing today.
- You are here:
- Home »
- Blog »
- Multiple Myeloma »
- Myeloma- Cardiomyopathy- Heart Failure
Myeloma- Cardiomyopathy- Heart Failure
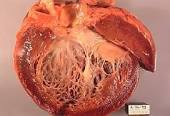
I am a long-term myeloma survivor. I underwent cardiotoxic chemotherapy regimens (vincristine, adriamycin/doxorubicin, cytoxan, busulphan, melphalan) during my conventional myeloma treatments between 1994-1997. I developed both chronic atrial fibrillation (Afib) and chemotherapy-induced cardiomyopathy (CIC) in late 2010.
The issue for myeloma patients and survivors to understand is if and when their chemotherapy regimens put them at risk for heart failure. The article below documents the lifelong progression of cardiotoxic chemotherapy.
According to my experience combined with the article linked below, the lifetime risk of chemotherapy-induced cardiomyopathy and risk of death increases for your entire life.
Further, I don’t think that conventional oncology fully understands the risks. This is just my opinion of course, but after reading my medical charts I got the distinct feeling that my oncologists knew that my chemotherapy regimens were cardiotoxic and my heart was being damaged but that was the risk that they were willing to take. With my life…
Five (5) courses of VAD infusion chemotherapy consisting of:
- Vincristine- 2 milligrams total dose over 4 days.-
- Adriamycin- 96 milligrams total dose over 4 days-
- Dexmethasone- 40 milligrams a day x 4 days-
Two (2) courses of high-dose cytoxan/cyclophophomide chemotherapy:
- Cytoxan- 9.45 grams intravenously over six hours-
- Prednisone- 200 milligrams a day for 4 days-
My autologous stem cell transplant chemotherapy regimens consisting of:
- Busulfan
- Melphalan
- Cytoxan-
My medical file contains a note written by Dr. Hillard Lazarus, the doctor in charge of my ASCT-
“Left ventricular compliance- The LVEF 7/20/95 = 77%. Dr. (Barry) Effron felt that the Echo findings were non-specific and the minor changes in the diastolic flow may have been due to previous adriamycin and there was no concern regarding high-dose chemotherapy…He (me, the patient) received high-dose cytoxan therapy on 10/18/95.”
I believe the article linked below documents me as a “missed opportunity.” University Hospitals of Cleveland and the physicians who treated me beginning with my cancer diagnosis in early 1994 are a prime example of what the article is talking about.
After exposing me to well-documented cardiotoxic chemotherapy regimens, instead of regularly monitoring me over the years, University Hospitals simply ignored my atrial fibrillation and reduced ejection fraction.
Not only did my standard-of-care multiple myeloma therapies not result in remission, the many therapies resulted in short, long-term and late stage side effects (CIC). To top it all off, U.H. charged me tens of thousands of dollars!
Have you been diagnosed with myeloma? Are you considering undergoing chemotherapy? To learn more about evidence-based, non-toxic, non-conventional therapies to reduce or prevent damage to your heart, kidneys, bones and bone marrow, scroll down the page, post a question or a comment and I will reply to you ASAP.
Thank you,
David Emerson
- MM Survivor
- MM Cancer Coach
- Director PeopleBeatingCancer
Recommended Reading:
- Exercise-based Cardiac Rehab- Chemotherapy-induced Cardiomyopathy
- Cardiac Rehabilitation- Omega 3 Fatty Acids, Heart Failure
- Can Heart Failure, Atrial Fibrillation Patients Have Sex?
Partially reversible bortezomib-induced cardiotoxicity: an unusual cause of acute cardiomyopathy
“Conclusion- In light of the above, cardiac function should be assessed at baseline with echocardiogram, BNP, and EKG, even in asymptomatic patients, as part of a routine workup before starting proteasome inhibitor therapy.
We also recommend that BNP be monitored serially during therapy or at the first hint of symptoms referable to cardiac failure. Repeat cardiac assessments after treatment should also be strongly considered. Further research is required to inform guidelines for cardiac assessment of patients on proteasome inhibitor therapy…”
Anthracycline
“Clinically the most important anthracyclines are doxorubicin, (adriamycin) daunorubicin, epirubicin and idarubicin.[4]…”
Heart Failure Progression During Cardiotoxic Chemotherapy
JACC: Heart Failure, Volume 6, Issue 12, December 2018, Pages 1023-1032
“Cardiotoxicity, often used to describe a decline in left ventricular ejection fraction (LVEF) related to potentially cardiotoxic chemotherapy, occurred in 9% of a cohort of 2,625 patients receiving anthracycline therapy, with the highest incidence observed within the first year after chemotherapy completion (1).
Regular monitoring of cardiac function with echocardiograms during and upon completion of chemotherapy facilitated early detection of cardiotoxicity, and intervention using guideline-directed heart failure (HF) medications, with subsequent improvement in LVEF…
Initiation of HF therapy is recommended as early as stage A (at risk, which includes patients receiving cardiotoxins) (Figure 1) and stage B (structural heart disease including a decline in LVEF), as defined by the American College of Cardiology and American Heart Association (2)…
In patients with cancer receiving potentially cardiotoxic chemotherapy, a 19% transition rate (22% transition for those receiving anthracyclines) from stage A to stage B HF was observed within 3 months, as documented by LVEF assessment by cardiac magnetic resonance (3).
The mean cumulative dose of doxorubicin in patients with stage B HF at 3 months and 6 months was <250 mg/m2, demonstrating that HF may occur at doses far below the “high dose” threshold of 250 mg/m2 (4), with subsequent need for cardiotoxicity monitoring regardless of doxorubicin dose...
Furthermore, the lack of correlation between change in LVEF and symptom scores supports the need for early consultation with cardio-oncologists for early detection and intervention of stage B HF before the irreversible progression to stage C HF with clinical symptoms.
The mistaken reliance on symptom development to guide the need for both the assessment of cardiac function and referral to cardio-oncologists has resulted in undetected transitions from stage A HF to stage B HF, which may progress to stage C (clinical HF) with early symptoms of HF overlooked or misdiagnosed/misinterpreted by non-cardiology specialists.
Once HF symptoms appear (stage C HF) in patients with reduced LVEF, the risk for progression to stage D HF increases, with a recent study of 964 HF with reduced EF patients, receiving care by cardiologists at an academic medical center, reporting an annualized 4.5% progression to stage D, despite guideline-directed therapy.
Whereas the 3-year progression rate to stage D was 12.2%, the 3-year combined mortality or progression rate was more than 2-fold, at 25.1%, that is,1 of 4 patients either dying or progressing to stage D, with earlier progression observed among nonischemic and African American patients (5)…
However, early detection of anthracycline-induced cardiotoxicity with early initiation of HF therapies resulted in partial to full recovery of LVEF (1), with lower incidence of adverse cardiac events observed in patients who recovered a normal LVEF, compared to those with persistent cardiac dysfunction, even if asymptomatic, thus underscoring the inadequacy of a symptom-guided approach.
Lack of cardio-oncology consultation may represent a missed opportunity for early diagnosis and intervention.”
American Heart Association Heart Failure Stages
- Stage A: Presence of heart failure risk factors but no heart disease and no symptoms
- Stage B: Heart disease is present but there are no symptoms (structural changes in heart before symptoms occur)
- Stage C: Structural heart disease is present AND symptoms have occurred
- Stage D: Presence of advanced heart disease with continued heart failure symptoms requiring aggressive medical therapy