Multiple myeloma and MRI scans go together like peas and carrots. For that matter so do X-rays, PET and CT scans. Unfortunately, gadolinium-based contrast agents (GBCAs), the contrast agent used for Magnetic Resonance Imaging (MRI) scans have been shown to promote multiple myeloma cell growth.
Knowing this, when I was about to have an MRI scan a few months ago, I simply asked the technician NOT to give me any “contrast agent.” This wasn’t a problem for either of us. Although, according to the study linked below, the lack of a contrast agent made the MRI scan more difficult to read.
So I think it is safe to say that multiple myeloma patients and survivors should avoid gadolinium-based contrast agents.
The article below, therefore, should be good news. Keep in mind however, that the new improved contrast agent is new and not tested when it comes to multiple myeloma. I, myself, will ask NOT to include contrast agents of any kind until I read studies documenting how the new contrast agent works for MM patients.
Have you been diagnosed with multiple myeloma? Are you experiencing any bone damage? Is one or more imaging studies like MRI in your future? Scroll down the page, post a question or comment and I will reply to you ASAP.
“Researchers at Massachusetts General Hospital (MGH) have taken a key step forward in developing a new, possibly safer contrast agent for use in magnetic resonance imaging (MRI) exams. Contrast enhanced MRI is a widely used diagnostic tool with over 30 million procedures performed annually. Currently, gadolinium-based contrast agents (GBCAs) are used for this purpose, but recently concerns have been raised about the long-term safety of the gadolinium metal ion…
Results of the study are available online at Investigative Radiology, and describe how a novel manganese-based agent (Mn-PyC3A) provides comparable tumor contrast enhancement to state of the art (GBCAs). The new agent also may be safer than GBCAs, because manganese from Mn-PyC3A is much more quickly and thoroughly cleared from the body than gadolinium from even the ‘safest’ GBCA.
A key feature of the new agent is that the manganese is tightly bound to a chelator which prevents it from interacting with cells or proteins in an adverse way and allows rapid elimination from the body after the imaging exam. “Without a chelator of sufficient strength, the manganese will be taken up by the liver and remain in the body…” With a strong chelator, the manganese is distributed throughout the body and can detect the location and size of lesions.
While the first GBCA was approved by the U.S. Food and Drug Administration (FDA) in 1988, there are lingering safety concerns about these agents.
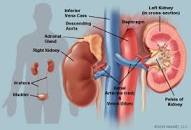
In 2007 it was determined that GBCAs can cause nephrogenic systemic fibrosis (NSF) when used in patients with kidney disease.
NSF is a rare,but devastating progressive condition that affects multiple organ systems. As a result, three GBCAs can no longer be used in patients with advanced kidney disease, while the use of other GBCAs is avoided. However, avoiding contrast enhanced MRI makes it harder to provide accurate diagnoses for these patients.
Also, it’s been known for several years that residual gadolinium can stay in the body for a very long time after an imaging procedure. Studies have demonstrated that gadolinium levels in the brain and other organs increase with increased exposure to GBCAs. Concerns around gadolinium retention caused the European Medicines Agency to remove several GBCAs from the market in Europe…
Derek Erstad, MD, clinical fellow in the Department of Surgery at MGH and lead author on the study points out that “A number of conditions require multiple follow up scans with GBCAs that result in increased gadolinium exposure. For instance, women with a high risk of breast cancer, brain cancer survivors, or patients with relapsing multiple sclerosis may receive frequent GBCA enhanced MRIs for surveillance…”
Multiple Myeloma Diagnostics- MRI w/Contrast Gadolinioum Adverse Events-
“in autopsies in 8 MM patients with repeated exposure to Omniscan, we quantified gadolinium in various tissues…We observed massive quantities of gadolinium accumulation in tissues of these MM patients regardless of their renal function…”
It’s unusual for multiple myeloma patients to develop adverse events aka side effects from multiple myeloma diagnostics- that is to say it is not the test that usually causes the adverse event, rather than chemotherapy, surgery or radiation. But, according to the articles linked below, Magnetic Resonance Imaging (MRI) testing with the contrast agent gadolinium, can cause a variety of adverse events.
According to research more than 80% of all MM survivors will develop bone events at some time while living with multiple myeloma. Further, as in my case, my personal experience as a newly diagnosed MM patient, it was my MRI, with contrast, that was able to diagnose very early evidence of MM. My pathologist referred to “uptake” within the bone lesion that indicated the presence of active multiple myeloma.
Speaking from personal experience then, it was an MRI with contrast that both diagnosed my MM AND caused my MM to spread.
Recommended Reading:
“Finally in autopsies in 8 MM patients with repeated exposure to Omniscan, we quantified gadolinium in various tissues using Inductively-coupled mass spectrometry. We observed massive quantities of gadolinium accumulation in tissues of these MM patients regardless of their renal function…”
“Gadolinium-based contrast agents (GBCAs) are central to MRI exams, providing critical information unavailable with other modalities. However, some patients experience acute adverse reactions and investigators of a recent study published in Radiology set out to get a clearer picture of these events.
“These acute adverse events are uncommon and most frequently occur within 1 hour of intravenous GBCA administration, with a reported incidence of seven to 240 per 10,000 injections…”
“This rarity has made it challenging to compare the rate of reactions between GBCAs and to determine what risk factors are associated with acute reactions.”
For their retrospective analysis, the team analyzed 281,945 intravenous MRI-related GBCA injections undertaken at a single institution. That added up to 158,000 patients with a median age of 55 years old.
They examined four GBCA’s, of those,
- 50% were gadodiamide,
- 33% were gadobutrol,
- 14% were gadobenate dimeglumine and
- 2.9% were gadoterate meglumine.
Reactions were classified as allergic-like or physiologic and as mild, moderate or severe according to ACR criteria.
After multivariate analysis, those who received gadobenate or gadobutrol had higher rates of allergic-like reactions than those who were administered gadodiamide.
“Gadobenate dimeglumine and gadobutrol are associated with higher rates of allergic-like reactions compared with gadodiamide and gadoterate meglumine, and gadoterate, gadobenate, and gadobutrol are associated with higher rates of physiologic reactions compared with gadodiamide, McDonald et al. wrote.
Of note, the severe reactions that occurred following gadobutrol administration were more serious and required longer hospitalizations than did the severe reactions that occurred following gadobenate administration. Two patients who received gadobutrol required subsequent extracorporeal membrane oxygenation support. No deaths related to GBCA administration were noted…”
“Conclusions– Gadolinium accumulation occurs in almost all patients undergoing contrast MRI screening after >5 MRIs. We advocate a screening protocol for patients with hereditary tumor syndromes that minimizes the Gadolinium dose. This can be accomplished by using a single administration to simultaneously screen for brain, spine and/or abdominal lesions, using an MRI protocol focused on either VHL- or TSC-specific lesions. Higher prevalence and rate of accumulation in VHL patients may be explained by the typical vascular leakage accompanying central nervous system hemangioblastomas…”