
Diagnosed with Cancer? Your two greatest challenges are understanding cancer and understanding possible side effects from chemo and radiation. Knowledge is Power!
Learn about conventional, complementary, and integrative therapies.
Dealing with treatment side effects? Learn about evidence-based therapies to alleviate your symptoms.
Click the orange button to the right to learn more.
Oncotype DX for Breast Cancer and DCIS- better than a second opinion?
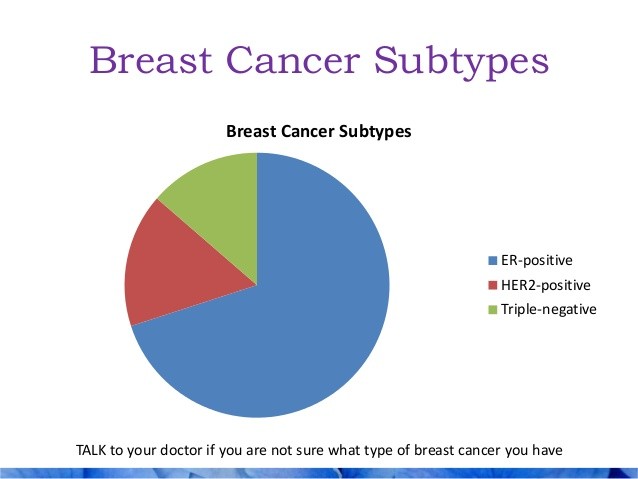
Ducal Carcinoma In-situ (DCIS) is Not Breast Cancer-Consider Evidence-based, Non-Toxic Therapies to Reduce Your Risk of Breast Cancer
The mission of PeopleBeatingCancer is to demystify cancer. I research and find studies, combine them with my experience, and blog about issues that I hope will explain complex ideas about cancer for cancer patients. Sometimes this is easier said than done.
I read an article yesterday that sparked a post about how pathologists may disagree over breast cancer biopsies. In defense of pathologists, I imagine that DCIS and atypia, both BC stage 0, are difficult to diagnose.
It is clear that any diagnosis beyond all clear or we found nothing on your mammogram or words to that effect, spark anxiety on the part of the patient. I have received many cancer coaching questions from women diagnosed with early breast cancers who ask about their prognosis and possible therapies.
If a woman is diagnosed with DCIS or atypia and has read my blog post highlighting the difficulty facing pathologists, the woman may have concerns about her health and experience the anxiety that comes with any breast cancer diagnosis. What are these tens of thousands of women supposed to do?
To learn more about DCIS and the evidence-based therapies that can help you prevent its spread into invasive breast cancer, please watch the video below:
To download the DCIS Guide, click here.
Enter the Oncotype DX Breast Cancer Assays. For more information about both conventional and non-conventional breast cancer therapies, scroll down the page, post a question or a comment and I will reply to you ASAP.
thank you,
David Emerson
- Cancer Survivor
- Cancer Coach
- Director PeopleBeatingCancer
Recommended Reading:
- Organic Diet-Fewer Cancer Dx’s, Longer Cancer Survival
- Testing Predicts DCIS Relapse
- Radiation for DCIS?
Breast Cancer Biopsy- More Art than Science?
“If you have had a breast cancer biopsy this year you are one of about 1.6 million people. The good news is that if you were diagnosed with invasive breast cancer, 98% of pathologists would agree on your diagnosis, according to the articles linked below.
The bad news is that if you were diagnosed with ductal carcinoma in situ (DCIS) or atypical ductal hyperplasia (atypia) your pathologist’s findings may cause disagreement with other pathologists.
Bottom line- get a second opinion.”
Initial Trends in the Use of the 21-Gene Recurrence Score Assay for Patients With Breast Cancer in the Medicare Population, 2005-2009
“Importance In 2006, the Centers for Medicare & Medicaid Services approved coverage for the use of the 21-gene recurrence score (RS) assay (Oncotype DX) in women with early-stage, estrogen receptor–positive, node-negative breast cancers to help guide recommendations for adjuvant chemotherapy...
Conclusions and Relevance The RS assay was adopted quickly in clinical practice after the Medicare coverage decision in 2006, and use appears to be consistent with guidelines and equitable across geographic and racial groups. Factors influencing adoption of the assay and its impact on adjuvant chemotherapy use in clinical practice remain important areas of study.”
Oncotype DX Recurrence Score in premenopausal women
“In the past 20 years, clinicians have shifted away from relying solely on clinicopathologic indicators toward increasing use of multigene expression assays in guiding treatment decisions regarding adjuvant chemotherapy for early-stage hormone receptor (HR)-positive, HER2-negative breast cancer. Oncotype DX Recurrence Score (RS) is one of the most widely used multigene assays when considering indications for adjuvant chemotherapy, and guidelines have recently incorporated its use in women with early HR-positive HER2-negative breast cancer and up to three positive lymph nodes.
While multiple retrospective and prospective clinical studies have demonstrated that most women with a low- to mid-range RS (0–25) can safely forgo chemotherapy, premenopausal women remain an important subgroup for which recommendations based on RS are ill-defined. The majority of patients included in clinical trials and retrospective analyses validating the use of RS have been postmenopausal women.
In the subgroup of premenopausal women with HR-positive HER2-negative breast cancer, studies indicate that traditional clinicopathologic methods for assessing risk continue to be powerful tools when combined with RS to predict benefit from chemotherapy. This suggests that there is an element of uncaptured risk inherent to the premenopausal state that evades characterization by RS alone.
This review describes the evidence that has supported the recommendation of RS in clinical guidelines, specifically focusing on data for its current use in premenopausal women. We review available data regarding the impact of the menstrual cycle on hormonally regulated gene expression, which may drive variations in the RS. Further research on the reliability and interpretation of the RS in the premenopausal subgroup is necessary and represents a gap in knowledge of how the RS should be applied in premenopausal women…”

