Leave a Comment:
13 comments
[…] Radiation for DCIS? […]
Reply[…] Radiation for DCIS? […]
ReplyHello,
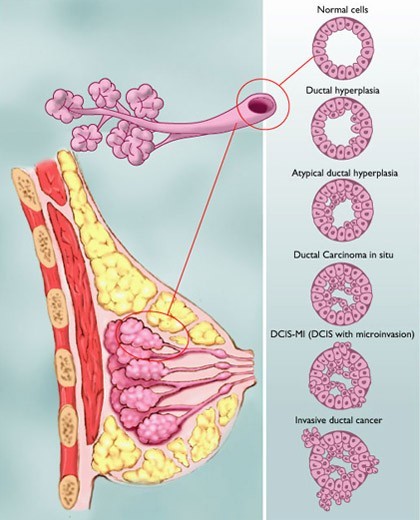
I just recently had surgery for DCIS. I’m contemplating having radiation. I have seen a lot of statistics and would like more information on DCIS grade 3. None of the articles seem to differentiate the odds between low to moderate and grade 3. I have completely changed my diet and added more supplements but am still unsure if 4 weeks of radiation is needed.
Hi Kim-
I sent the DCIS guide to you via your email address.
Good luck.
David Emerson
Reply[…] Radiation for DCIS? […]
Reply[…] Radiation for DCIS? […]
Reply[…] Radiation for DCIS? […]
ReplyHi David,
I have been recently diagnosed with DCIS. I’m interested in having lumpectomy and anti-hormonal treatment without radiation. Interested in your opinion.
Hi Sue-
Several things. Keep in mind that DCIS is pre-cancer, not cancer. Yes, this diagnosis is difficult and scary but try to keep it in perspective. Secondly, while my role is to give you evidence-based information and research, and yes, my thinking, only you can decide what is best for you.
The therapies you list- lumpectomy, anti-hormonal therapy and radiation, all have been developed in an effort to reduce the patient’s (you) risk of relapse or in this case, a diagnosis of full blown breast cancer. The real issue, in my view, it less about “relapse” and more about dieing of breast cancer. The short, long-term and late stage side effects of radiation therapy, in my opinion, are under reported and should be studied more. But this is just my feeling…
The next issue then, is to further reduce the risk of breast cancer. I encourage you to consider taking the Oncotype DX DCIS test to learn more about your risk of relapse.
The next step is to learn about and adopt those evidence-based, non-toxic therapies shown to reduce the risk of BC. Nutrition, supplementation, exercise, etc. all have been shown to reduce the risk of BC. As a cancer survivor myself, I subscribe to the practice of non-toxic therapies shown to reduce the risk of one’s cancer relapse.
As an aside, supplements such as curcumin and omega 3 fatty acids have been shown to both reduce the risk of BC as well as reduce the syptomsof therapies like tamoxifen.
Please read this article as I think it phrases the issues-click now-
All to say that if you choose not to undergo radiation therapy, and follow the above, you can reduce your risk of relapse to a tiny fraction and may experience much higher quality of life.
My apologies for being so long-winded above. Let me know if you have questions or if you want me to provide specific therapies for you to consider.
thanks and hang in there,
David Emerson
Reply[…] Radiation for DCIS? […]
Reply[…] Radiation for DCIS? […]
ReplyWhat are some non-toxic therapies shown to reduce the risk of DCIS relapse?
ReplyHi Jean-
The info linked below is only a partial list of non-toxic therapies. My cancer is very different (blood cancer called multiple myeloma) but I have taken many of the therapies below for years and I remain in complete remission.
If you have been diagnosed with DCIS and you have undergone a lumpectomy to remove the abnormal cells, you may be thinking through your next therapy steps to consider your options.
Whatever course you choose please consider the Oncotype DX DCIS test- this will tell you your risk of relapse-
Further, please consider lifestyle therapies such as acupuncture.
Further, a growing number of studies cite moderate but frequent exercise as BC therapy.
Further, supplements such as
1) iodine
2) curcumin-
4) cruciferous veggies- the research is thin here but I’ve read other studies that consider cruciferous veggies to be anti-cancer.
I am a cancer coach. I can design a complete therapy plan if you are interested.
David Emerson
Reply