“Trials of lenalidomide (revlimid) in a less intensive approach has shown promise in delaying disease progression and should be investigated further in clinical trials.”
You’ve been diagnosed with Smoldering Multiple Myeloma (SMM) and your oncologist has been talking to you about entering a clinical trial to see if you can slow the progression of your SMM to full MM.
But you’re just not sure when to begin treatment. I mean, SMM is not cancer. And let’s face it- you don’t want to undergo any chemo… of any kind.
Your goals are:
- Slow my SMM progression to full MM-
- Achieve as long an overall survival (OS) aka length of life, as possible-
- Achieve as high a quality of life as possible-
What does the research about treating smoldering multiple myeloma say?
The top study linked and excerpted below was conducted at the end of 2020. All of the studies, all of the therapies used, none achieved a longer overall survival. All treatments led to adverse events aka side effects. Except one.
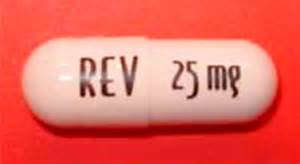
Low dose Revlimid aka lenalidomide. The least toxic regimen is the only treatment, according to the research below, that yielded “superiority in delaying disease” as well as “superior overall survival over observation across the trials.”
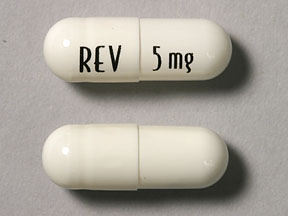
Unfortunately, the studies don’t mention specific dosages of lenalidomide used in the studies. The standard of care dose for newly diagnosed MM patients is 25mg. The dose used for “low dose maintenance therapy” is usually 10mg.
Just my opinion but keep in mind that Smoldering Multiple Myeloma is, by definition, a tiny amount of MM in your system- plasma cells in your bone marrow. Undergoing full doses of Revlimid will increase your risk of side effects. Consider low doses of Revlimid such as 2.5 mg or 5 mg.
Lastly, the second and third studies linked below about about curcumin. In hundreds of studies, curcumin has been shown to kill MM. According to research, curcumin can enhance the efficacy of Revlimid while reducing toxicity.
Further, a number of evidence-based but non-toxic therapies such as
- nutrition,
- supplementation and
- lifestyle therapies
have shown the ability to reduce the risk of a multiple myeloma diagnosis.
Consider combining the best of conventional therapies with the best of evidence-based but non-conventional MM therapies to manage your smoldering multiple myeloma.
To learn more about managing both pre-MM and full MM, scroll down the page, post a question or comment and I will reply to you ASAP.
Thank you,
David Emerson
- MM Survivor
- MM Cancer Coach
- Director PeopleBeatingCancer
Recommended Reading:
The effect of intervention versus watchful waiting on disease progression and overall survival in smoldering multiple myeloma: a systematic review of randomized controlled trials
“The question of who, when, and what to use for the treatment of SMM remains equivocal. We performed a systematic review of randomized controlled trials and summarized the current evidence supporting the best approach to the management of SMM…
A comprehensive literature search of Medline/PubMed, PubMed Central, Embase, Scopus, Web of Science, Wiley Cochrane Library, CINAHL, clinicaltrial.gov, and conference proceedings of ASCO, ASH, EHA, and ESMO was performed on October 25, 2020. Synthesis of the result was done using narrative analysis…
Of the total 1560 identified records, 10 eligible studies involving 1157 patients made up of 580 in the intervention group and 577 in the control group were included in this review.
- Three early trials of melphalan and prednisone fail to demonstrate any significant impact on disease progression with major toxicities reported.
- Three trials on bisphosphonate monotherapy show reduced skeletal-related events without any clinical effect on disease progression.
- Lenalidomide monotherapy or as part of a combination therapy demonstrates superiority in delaying disease progression over observation. Only Lenalidomide and dexamethasone combination demonstrated superior overall survival over observation across the trials.
CONCLUSION: Trials of lenalidomide in a less intensive approach has shown promise in delaying disease progression and should be investigated further in clinical trials.”
“Background: Curcumin, the active component of the Curcuma longa plant, has been shown to potentiate the effect of the immunomodulatory drugs (IMiDs) thalidomide and Bortezomib against human myeloma cell lines and a nude mice model. Its effect on the other IMid, lenalidomide, has not been evaluated. This study aims to investigate the mechanism of action of curcumin and its potential ability to positively interact with lenalidomide.
Method: we designed an in-vitro study to investigate the cytotoxic and chemo-sensitising effects of curcumin alone and in combination with lenalidomide on the human myeloma H929 cell line.
Results: Incubation of H929 cells with curcumin (30mM) or lenalidomide (2.5 mM) for 3 days resulted in 26.35% (±1.06) and 30.81%(±2.98) apoptotic cells respectively. When 30 mM curcumin was combined with 2.5 mM lenalidomide, 50.4% (±3.37) apoptotic cells were detected by flow cytometry and the increase was significant compared to either curcumin alone or lenalidomide alone (anova p = 0.0026). Furthermore, gene analysis studies show that curcumin enhances the cytotoxic effect of lenalidomide via suppression of the cereblon and multi-drug resistant genes.
Conclusion: Curcumin exerts a cytotoxic effect additive to that of lenalidomide on H929 myeloma cells, and it also enhances the chemo-sensitizing effects of this agent.”
Extensive research over the past half century has shown that curcumin (diferuloylmethane), a component of the golden spice turmeric (Curcuma longa), can modulate multiple cell signaling pathways. Extensive clinical trials over the past quarter century have addressed the pharmacokinetics, safety, and efficacy of this nutraceutical against numerous diseases in humans. Some promising effects have been observed in patients with various pro-inflammatory diseases including cancer, cardiovascular disease, arthritis, uveitis, ulcerative proctitis, Crohn’s disease, ulcerative colitis, irritable bowel disease, tropical pancreatitis, peptic ulcer, gastric ulcer, idiopathic orbital inflammatory pseudotumor, oral lichen planus, gastric inflammation, vitiligo, psoriasis, acute coronary syndrome, atherosclerosis, diabetes, diabetic nephropathy, diabetic microangiopathy, lupus nephritis, renal conditions, acquired immunodeficiency syndrome, β-thalassemia, biliary dyskinesia, Dejerine-Sottas disease, cholecystitis, and chronic bacterial prostatitis. Curcumin has also shown protection against hepatic conditions, chronic arsenic exposure, and alcohol intoxication. Dose-escalating studies have indicated the safety of curcumin at doses as high as 12 g/day over 3 months.