
Diagnosed with Cancer? Your two greatest challenges are understanding cancer and understanding possible side effects from chemo and radiation. Knowledge is Power!
Learn about conventional, complementary, and integrative therapies.
Dealing with treatment side effects? Learn about evidence-based therapies to alleviate your symptoms.
Click the orange button to the right to learn more.
- You are here:
- Home »
- Blog »
- side effects ID and prevention »
- Dexrozoxane for Cardioprotection?!?
Dexrozoxane for Cardioprotection?!?
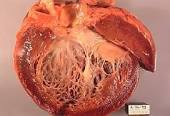
Dexrozoxane, according to the research below, is used to prevent heart damage to cancer patients who undergo cardiotoxic chemotherapies such as doxorubicin.
Unfortunately, dexrozoxane causes a host of short and long-term side effects as well- according to the research linked below.
In my experience, both conventional oncology as well as cardiology will always prescribe FDA approved, “safe and effective” therapies such as dexrozoxane rather than evidence-based non-conventional therapies. Both dex. and dox. are neither safe nor effective if the cancer patient develops cardiomyopathy.
I don’t think anyone should criticize a cancer therapy without offering an alternative. In this case, CoQ10, according to research, also prevents heart damage from cardiotoxic chemo regimens as well.
I have a firsthand perspective of doxorubicin (anthracyclines) causing cardio toxicity because in 1995, I underwent doxorubicin, as well as several other cardiotoxic chemo regimens, and developed chemotherapy-induced cardiomyopathy (CIC)in late 2010-fully 15 years after my chemo.
I have stabilized my CIC by following a heart-healthy lifestyle including
- Mediterranean Diet
- Frequent, moderate exercise
- Nutritional supplementation including CoQ10-see below
- Heart healthy lifestyle therapies
I supplement with CoQ10 daily (as well as several other heart healthy nutritional supplements), I no longer drink alcohol, I to all the therapies above- in short, I try to walk the walk and talk the talk.
If you are a pediatric or AYA cancer patient and you are considering cardiotoxic chemo regimens, please consider evidence-based, non-conventional therapies to protect your heart.
To learn more, scroll down the page, post a question or a comment and I will reply to you ASAP.
Thank you,
David Emerson
- Cancer Survivor
- Cancer Coach
- Director PeopleBeatingCancer
Cardioprotection for 20 Years for Children With Cancer
“Dexrazoxane (Totect, Zinecard) provides long-term cardioprotection for children treated with doxorubicin for cancer, and its co-administration with doxorubicin should be standard of care, say researchers in a report publishedpublished January 20 in the Journal of Clinical Oncology…
Underused in Children
Despite recent progress in cancer therapeutics, doxorubicin and other anthracyclines remain cornerstones of pediatric cancer treatment. However, they come with a risk of late-onset cardiomyopathy that can approach 25% in some groups by the time survivors are 40 years old, and the risk has not declined appreciably in 50 years, the authors point out.
Outside of using lower, possibly subtherapeutic doses of anthracyclines, dexrazoxane is the only cardioprotective option available to children with cancer…
Coenzyme Q10 protects against doxorubicin-induced cardiomyopathy via antioxidant and anti-apoptotic pathway
“Doxorubicin (Dox) is an anthracycline antibiotic that treats a variety of malignancies. Unfortunately, its cardiotoxicity limits its therapeutic usefulness. Coenzyme Q10 (CoQ10) has effectively treated and prevented various cardiac diseases and toxicities. This study aimed to evaluate the possible antioxidative and anti-apoptotic cardioprotective effects of CoQ10 against doxorubicin-induced histopathological and molecular changes in cardiomyocytes…
In conclusion, CoQ10 protects against Dox-induced cardiotoxicity through the regulation of proapoptotic and anti-apoptotic gene expression…”
Dexrazoxane for the treatment of chemotherapy-related side effects
“Presently, dexrazoxane is licensed in many parts of the world for two different indications: prevention of cardiotoxicity from anthracycline-based chemotherapy, and prevention of tissue injuries after extravasation of anthracyclines…
The most serious side effect of repeated anthracycline therapy is late cardiomyopathy. The clinical presentation is that of congestive heart failure, and the major risk factor is the cumulative dose of the anthracycline…
Aggravating risk factors for developing cardiomyopathy after anthracycline therapy include older age, radiation therapy to the mediastinal region, and combination chemotherapy with anticancer agents such as cyclophosphamide, trastuzumab, and taxanes. The clinical problem is of major importance in the treatment of pediatric patients.
Dexrazoxane injection may cause side effects. Tell your doctor if any of these symptoms are severe or do not go away:
- pain or swelling in the place where the medication was injected
- nausea
- vomiting
- diarrhea
- constipation
- stomach pain
- loss of appetite
- dizziness
- headache
- excessive tiredness
- difficulty falling asleep or staying asleep
- depression
- swelling of the arms, hands, feet, ankles, or lower legs
Some side effects can be serious. If you experience any of these symptoms, call your doctor immediately or get emergency medical treatment:
- sore throat, fever, chills, cough, and other signs of infection
- unusual bruising or bleeding
- pale skin
- weakness
- shortness of breath
- rash
- itching
- hives
- difficulty breathing or swallowing
- swelling of the eyes, face, mouth, lips, tongue, or throat
- dizziness
- fainting
Some people who took a medication that is very similar to dexrazoxane injection developed new forms of cancer. There is not enough information to tell if receiving dexrazoxane injection increases the risk that you will develop a new type of cancer. Talk to your doctor about the risks of receiving this medication.
Dexrazoxane injection may cause other side effects. Call your doctor if you have any unusual problems while receiving this medication.
If you experience a serious side effect, you or your doctor may send a report to the Food and Drug Administration’s (FDA) MedWatch Adverse Event Reporting program online (http://www.fda.gov/Safety/MedWatch) or by phone (1-800-332-1088).
In case of emergency/overdose
In case of overdose, call the poison control helpline at 1-800-222-1222. Information is also available online at https://www.poisonhelp.org/help. If the victim has collapsed, had a seizure, has trouble breathing, or can’t be awakened, immediately call emergency services at 911.
Symptoms of overdose may include:
- sore throat, fever, chills, and other signs of infection
- unusual bruising or bleeding
- pale skin
- shortness of breath
- excessive tiredness



