Lasix reduces fluid yet also causes a host of side effects. The study linked below questions the need for Lasix (furosemide) in patients with stable heart failure.
Lasix (furosemide) main use is to cause heart failure patients to urinate in an effort to reduce excessive fluid accumulation (edema) caused by congestive heart failure. The trouble is, lasix also causes a host of side effects.
Full disclosure: A diagnosis of multiple myeloma in early 1994 led to aggressive chemotherapy, radiation and an autologous stem cell transplant. The long-term and late stage side effects that I have sustained is extensive. I reached end-stage MM in September of 1997.
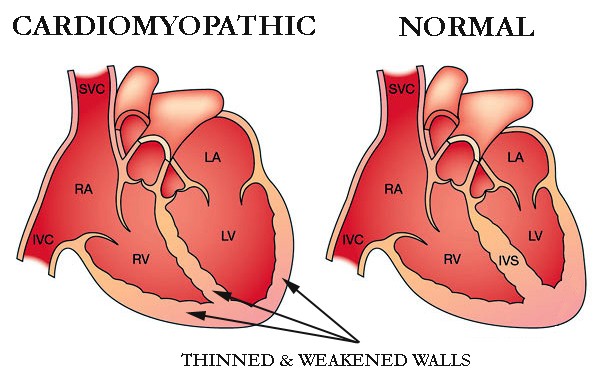
I flipped into chronic atrial fibrillation in late 2010 and was diagnosed with chemotherapy-induced cardiomyopathy.
Chances are, if you have some form of heart failure, you are going to be taking a number of therapies (pills). All medical therapies cause side effects. Therefore, you have to wonder if a given therapy is worth the side effects.
In my experience, medical professionals often blow off the seriousness of side effects. Nothing against M.D.’s but I have never had a relationship with a doctor (oncologist or cardiologist) who worried about what a prescribed medication might do to me. Never.
I’m not telling you to not take lasix. I’m telling you to read the study linked below, learn about the lasix/furosemide side effects and ask your cardiologist if this medication is really necessary in your case?
According to the article below, if your heart faillure is stable, consider stopping furosemide. Consider evidence-based, non-toxic therapies to enhance your cardiac rehabilitation.
Do you have heart failure? Scroll down the page, post a question or comment and I will reply to you ASAP.
Thank you,
David Emerson
- Cancer Survivor
- Cancer Coach
- Director The Galen Foundation
Recommended Reading:
“Lasix (furosemide) is an anthranilic acid derivative that is used as a strong diuretic in adults and children to treat excessive fluid accumulation (edema) caused by congestive heart failure…
Side effects of Lasix include:
Serious side effects of Lasix include:
“…a scientific congress of the European Society of Cardiology (ESC) in Athens (26 May 2019), looked at the safety and tolerability of deprescribing the diuretic furosemide in 188 patients with chronic but stable heart failure — half of whom would continue taking furosemide while the other half were given a placebo.
During the 90-day double-blind randomised trial, the researchers assessed patients’ levels of dyspnoea (shortness of breath) four times, and tracked the number of patients who requested additional diuretics to what they were already being given. They found no difference between the two groups in terms of self-perception of their dyspnoea during the 90 days.
It was found that 72 patients (75.3%) in the placebo group and 78 patients (83.9%) in the furosemide group were free of furosemide reuse during follow-up (p=0.16).
In the placebo group, there were five heart failure hospitalisations or emergency room visits, while in the furosemide group, two patients died and there were three heart failure hospitalisations or emergency room visits.
The team concluded that stopping diuretic use appeared “to be well tolerated in most of the patients and might deserve consideration in the appropriate clinical setting”.”