Leave a Comment:
3 comments
[…] A-fib- Reduce Risk of Stroke, Dementia Now- […]
Reply[…] A-fib- Reduce Risk of Stroke, Dementia Now- […]
Reply[…] A-fib- Reduce Risk of Stroke, Dementia Now- […]
Reply
Learn about conventional, complementary, and integrative therapies.
Dealing with treatment side effects? Learn about evidence-based therapies to alleviate your symptoms.
Click the orange button to the right to learn more.
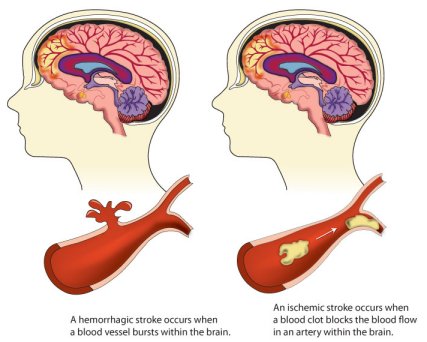
The researchers found that people with A-fib showed a faster rate of decline in thinking and memory skills than those without the condition, and were 40% more likely to develop dementia…
The study finding make sense. Slower blood flow increases risk to those bodily systems that depend on blood flow. Your brain depends on blood flow. A-fib slows blood flow. My ejection fraction is 45%.
I was diagnosed with multiple myeloma in 2/94, underwent five courses of VAD induction therapy from April-August of ’95, two courses high-dose cytoxan therapy in September of ’95 and finished off the year with an autologous stem cell transplant featuring busulfan and melphalan.
I developed chronic A-fib in the fall of 2010. The long-term solution then, according to the article below, is to undergo therapies that 1) reduce my risk of stroke and 2) reduce my risk of dementia. Because I am a long-term cancer survivor who’s body has been damaged by short, long-term and late stage side effects of conventional medicine (think chronic a-fib), I would like to undergo therapies that have no side effects.
Okay, I understand that all therapies have some side effects. Exercising too much is bad. I’m talking about, for example, exercising enough to reduce my risk of stroke and dementia, but not kill myself doing so.
According to my research, the therapies below are evidence-based, non-toxic methods of reducing my risk of stroke and dementia.
There are many other evidence-based, non-toxic therapies shown to reduce my risk of stroke and dementia now that I’m living with chronic a-fib but I thought the five therapies above were enough for this blog post.
Are you living with atrial fibrillation? Would you like to reduce your risk of stroke and dementia? Scroll down the page, post a question or comment and I will reply to you ASAP.
Thank you,
David Emerson
“People with atrial fibrillation, or a-fib, a type of irregular heartbeat that is linked with stroke, also may have a greater risk of dementia than those without the condition, according to a study published online Oct. 10, 2018, by Neurology.
Researchers recorded medical data from 2,685 people, average age 73. None of them had dementia and 243 had a-fib. After nine years, another 279 people developed a-fib. The researchers found that people with a-fib showed a faster rate of decline in thinking and memory skills than those without the condition, and were 40% more likely to develop dementia.
It’s well known that people with A-fib can significantly lower their risk of stroke by taking anticoagulant drugs (blood thinners), and the study found that people who took the drugs also had a 60% lower dementia risk. (The researchers found that aspirin did not have the same effect.)
The thinking is that blood thinners stop the formation of small clots that can cause small unnoticed strokes, which could lead to faster cognitive decline and a higher risk of dementia.
“Based on a review of these studies, it is evident that better bioavailability of formulated curcumin (CU) products is mostly attributed to improved solubility, stability, and possibly low first-pass metabolism”
A search of the Pubmed database for the word curcumin yields 601 studies spaning health topics from multiple myeloma and colorectal cancer, to chemotherapies that synergizes with CU, to Alzheimer’s Disease, arthritis and more. Based on years of reading studies and personal accounts, I think it is safe to say that CU supplementation is safe and relatively inexpensive.
I have read about myeloma patients taking daily doses of CU from 400 milligrams to 8 grams (1000 milligrams = 1 gram). By almost any measure, CU is a safe, inexpensive wonder drug.
The only challenge is that CU is famously difficult to absorb in the body. In other words, a person has to mix curcumin with some sort of fat (coconut oil, chocolate, etc.) or take a brand of curcumin capsule that is already formulated to be more “bioavailable” in order to derive the full benefit of CU.
The study linked and exerpted below reviews different formulations of CU. The study itself lists the three most bioavailable formulation/brand of CU and I’ve added an excerpt from a further review from Consumerlab.com that lists four additional bioavailable brands of CU.
I consult the independent evaluation service Consumerlab.com frequently. For one low annual payment, I can read about and evaluate all of the nutritional supplement that I take.
“CU is a bright yellow chemical produced by some plants. It is the principal curcuminoid of turmeric (Curcuma longa), a member of the ginger family, Zingiberaceae. It is sold as an herbal supplement, cosmetics ingredient, food flavoring, and food coloring.[1]“
“Curcumin is a widely studied natural compound which has shown tremendous in vitro therapeutic potential. Despite that, the clinical efficacy of the native CU is weak due to its low bioavailability and high metabolism in the gastrointestinal tract. During the last decade, researchers have come up with different formulations with a focus on improving the bioavailability of curcumin. As a result, a significant number of bioavailable curcumin-based formulations were introduced with the varying range of enhanced bioavailability.
The purpose of this review is to collate the published clinical studies of CU products with improved bioavailability over conventional (unformulated) CU. Based on the literature search, 11 curcumin formulations with available human bioavailability and pharmacokinetics data were included in this review. Further, the data on clinical study design, analytical method, pharmacokinetic parameters and other relevant details of each formulation were extracted.
Based on a review of these studies, it is evident that better bioavailability of formulated curcumin products is mostly attributed to improved solubility, stability, and possibly low first-pass metabolism. The review hopes to provide a quick reference guide for anyone looking information on these bioavailable curcumin formulations.
Based on the published reports,
exhibited over 100-fold higher bioavailability relative to reference unformulated CU. Suggested mechanisms accounting for improved bioavailability of the formulations and details on the bioanalysis methods are also discussed.”
According to Consumerlab.com:
“Novasol has the highest bioavailability (185 x compared to unforumulated CU), followed by Curcuwin (136 x), Longvida (100 x), Meriva (48 x), BCM-95 (27 x), Curcumin C3 Complex + Bioperene (20 x), and then Theracumin (16 x).”
[…] A-fib- Reduce Risk of Stroke, Dementia Now- […]
Reply[…] A-fib- Reduce Risk of Stroke, Dementia Now- […]
Reply[…] A-fib- Reduce Risk of Stroke, Dementia Now- […]
Reply