“Overall, our results suggest that curcumin potentiates the antitumor effect of BCG through the inhibition of NF-kappaB and induction of TRAIL receptors in bladder cancer cells…”
According to the study linked and excerpted below “Up to 40% of patients with non-muscle-invasive bladder cancer (NMIBC) will fail intravesical bacillus Calmette-Guérin (BCG) therapy…” Further, according to the other study, curcumin both potentiates the efficacy of BCG as well as kills bladder cancer.
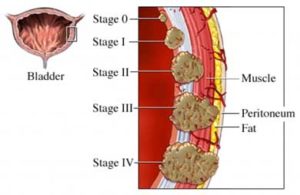
As a bladder cancer patient, your challenge is to find those therapies that offer you the best chance of success. In this case your success may be with taking an integrative approach by combining BCG therapy and curcumin.
I am a cancer survivor and cancer coach. I have supplemented with Life Extension Super Bio-Curcumin for more than 10 years and I have remained in complete remission from my “incurable” cancer all that time.
Please scroll down, post a question or comment and I will reply to you ASAP.
Thank you,
David Emerson
- Cancer Survivor
- Cancer Coach
- Director PeopleBeatingCancer
Recommended Reading:
“Overall, our results suggest that curcumin potentiates the antitumor effect of BCG through the inhibition of NF-kappaB and induction of TRAIL receptors in bladder cancer cells…”
Up to 40% of patients with non-muscle-invasive bladder cancer (NMIBC) will fail intravesical bacillus Calmette-Guérin (BCG) therapy. There is unfortunately no current gold standard for salvage intravesical therapy after appropriate BCG treatment. Indeed, outcomes are at best suboptimal. The vast majority of low-grade NMIBC are prone to recur but very rarely progress.
Failure after intravesical BCG in these patients is usually superficial and low-grade. At the other end of the spectrum, failure to respond to BCG in high-risk T1 bladder cancer and/or carcinoma in situ (CIS or TIS) is more problematic, since those tumours often have the potential to progress to muscle invasion.
In these cases, radical cystectomy remains the mainstay after BCG failure. With appropriate selection, certain patients who “fail” BCG (but with favourable risk factors) can be managed with intravesical regimens, including repeated BCG, BCG plus cytokines, intravesical chemotherapy, thermochemotherapy or new immunotherapeutic modalities. In this review, reasons explaining BCG failure, how to define BCG failure, optimal risk stratification and prediction of response and management of BCG failures are discussed.
The Most BioAvailable Curcumin Formulas
“Based on a review of these studies, it is evident that better bioavailability of formulated curcumin (CU) products is mostly attributed to improved solubility, stability, and possibly low first-pass metabolism”
A search of the Pubmed database for the word curcumin yields 601 studies spaning health topics from multiple myeloma and colorectal cancer, to chemotherapies that synergizes with CU, to Alzheimer’s Disease, arthritis and more. Based on years of reading studies and personal accounts, I think it is safe to say that CU supplementation is safe and relatively inexpensive.

I have read about myeloma patients taking daily doses of CU from 400 milligrams to 8 grams (1000 milligrams = 1 gram). By almost any measure, CU is a safe, inexpensive wonder drug.
The only challenge is that CU is famously difficult to absorb in the body. In other words, a person has to mix curcumin with some sort of fat (coconut oil, chocolate, etc.) or take a brand of curcumin capsule that is already formulated to be more “bioavailable” in order to derive the full benefit of CU.
The study linked and exerpted below reviews different formulations of CU. The study itself lists the three most bioavailable formulation/brand of CU and I’ve added an excerpt from a further review from Consumerlab.com that lists four additional bioavailable brands of CU.
Recommended Reading:
“CU is a bright yellow chemical produced by some plants. It is the principal curcuminoid of turmeric (Curcuma longa), a member of the ginger family, Zingiberaceae. It is sold as an herbal supplement, cosmetics ingredient, food flavoring, and food coloring.[1]“
“Curcumin is a widely studied natural compound which has shown tremendous in vitro therapeutic potential. Despite that, the clinical efficacy of the native CU is weak due to its low bioavailability and high metabolism in the gastrointestinal tract. During the last decade, researchers have come up with different formulations with a focus on improving the bioavailability of curcumin. As a result, a significant number of bioavailable curcumin-based formulations were introduced with the varying range of enhanced bioavailability.
The purpose of this review is to collate the published clinical studies of CU products with improved bioavailability over conventional (unformulated) CU. Based on the literature search, 11 curcumin formulations with available human bioavailability and pharmacokinetics data were included in this review. Further, the data on clinical study design, analytical method, pharmacokinetic parameters and other relevant details of each formulation were extracted.
Based on a review of these studies, it is evident that better bioavailability of formulated curcumin products is mostly attributed to improved solubility, stability, and possibly low first-pass metabolism. The review hopes to provide a quick reference guide for anyone looking information on these bioavailable curcumin formulations.
Based on the published reports,
exhibited over 100-fold higher bioavailability relative to reference unformulated CU. Suggested mechanisms accounting for improved bioavailability of the formulations and details on the bioanalysis methods are also discussed.”
According to Consumerlab.com:
“Novasol has the highest bioavailability (185 x compared to unforumulated CU), followed by Curcuwin (136 x), Longvida (100 x), Meriva (48 x), BCM-95 (27 x), Curcumin C3 Complex + Bioperene (20 x), and then Theracumin (16 x).”
APPIP ERROR: amazonproducts[
AccessDeniedAwsUsers|The Access Key Id AKIAJAJ37JVNL7OUU4CA is not enabled for accessing this version of Product Advertising API. Please migrate your credentials as referred here https://webservices.amazon.com/paapi5/documentation/migrating-your-product-advertising-api-account-from-your-aws-account.html.
]




