The studies linked excerpted below focus on ANP and brain cancer. The results of these studies indicate that ANP is efficacious in the treatment of both adult and pediatric brain cancer.
Dr. Stanislaw Burzynski, the Burzynski Research Institute and antineoplaston therapy (ANP) is one of the most controversial doctors/cancer therapies today. Dr. Burzynski has been called everything from a savior to a quack.
If you have been diagnosed with brain cancer then you probably know that conventional oncology has little to offer you that is an effective therapy that does not include the possibility of serious collateral damage.

After three years of aggressive conventional therapy including high-dose chemotherapy and an autologus bone marrow transplant, two relapses and finally “there is nothing more we can do for you” from my oncologist, from 2/94-9/97, I traveled to Houston, Texas and began intravenous ANP in November of 1997. After 10 months on intravenous ANP and another 7 on capsules-A10, A2-1, I achieved complete remission from my multiple myeloma where I remain today.
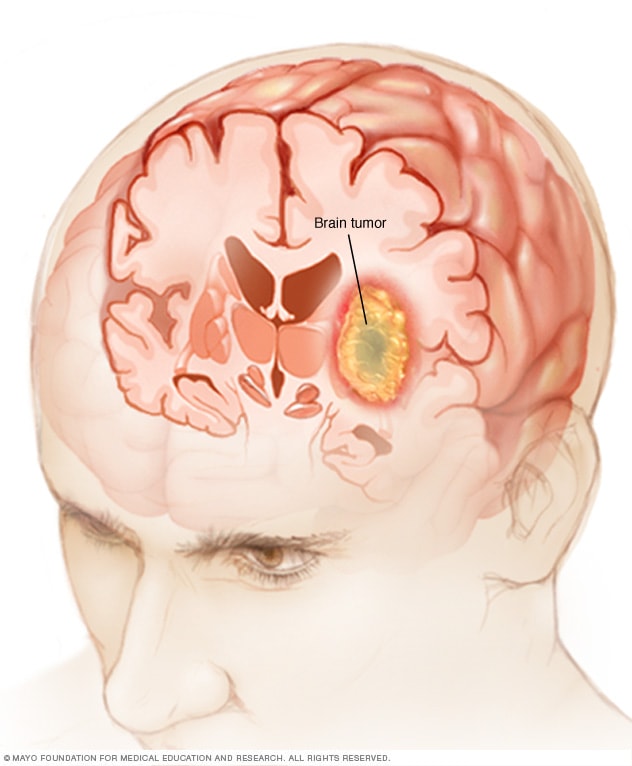
The studies linked excerpted below focus on ANP and brain cancers. The BRI and ANP therapy does not promise a cure. This therapy promises an effective alternative to conventional (FDA approved) brain cancer therapy. The results of these studies indicate that ANP is efficacious in the treatment of both adult and pediatric brain cancer.
I am both a cancer survivor and cancer coach. Have you been diagnosed with brain cancer? What type? What stage? To learn more about both conventional and non-conventional therapies for you please scroll down the page, post a question or comment and I will reply to you ASAP.
Thanks and hang in there,
David Emerson
- Cancer Survivor
- Cancer Coach
- Director PeopleBeatingCancer
Recommended Reading:
Brainstem glioma carries the worst prognosis of all malignancies of the brain. Most patients with brainstem glioma fail standard radiation therapy and chemotherapy and do not survive longer than 2 years. Treatment is even more challenging when an inoperable tumor is of high-grade pathology (HBSG)..
The overall survival at 2 and 5 years was 39% and 22%, respectively, and maximum survival was more than 17 years for a patient with anaplastic astrocytoma and more than 5 years for a patient with glioblastoma. Progression-free survival at 6 months was 39%. Complete response was achieved in 11%, partial response in 11%, stable disease in 39%, and progressive disease in 39% of patients. Antineoplastons were tolerated very well with 1 case of grade 4 toxicity (reversible anemia).
“Primitive neuroectodermal tumors (PNETs) are usually successfully treated with craniospinal radiation and chemotherapy; however, difficulties with standard treatment can be encountered in very young children, in adult patients at high risk of complication from standard treatment, and in patients with recurrent tumors. Thirteen children, either with recurrent disease or high risk, were treated in phase II studies with antineoplastons (ANP).
The median age of patients was 5 years, 7 months (range, 1-11). Medulloblastoma was diagnosed in 8 patients, pineoblastoma in 3 patients, and other PNET in 2 patients. Previous treatments included surgery in 12 patients (1 had biopsy only, suboccipital craniotomy), chemotherapy in 6 patients, and radiation therapy in 6 patients. Six patients had not received prior chemotherapy or radiation.
The treatment consisted of intravenous infusions of 2 formulations of ANP, A10 and AS2-1, and was administered for an average of 20 months. The average dosage of A10 was 10.3 g/kg/d and of AS2-1 was 0.38 g/kg/d. Complete response was accomplished in 23%, partial response in 8%, stable disease in 31%, and progressive disease in 38% of cases. Six patients (46%) survived more than 5 years from initiation of ANP; 5 were not treated earlier with radiation therapy or chemotherapy. The serious side effects included single occurrences of fever, granulocytopenia, and anemia. The study is ongoing and accruing additional patients.
The percentage of patients’ response is lower than for standard treatment of favorable PNET, but long-term survival in poor-risk cases and reduced toxicity makes ANP promising for very young children, patients at high risk of complication of standard therapy, and patients with recurrent tumors.”
“Complete response was accomplished in 33%, partial response in 25%, and stable disease in 33% of patients, and there was no progressive disease. One patient was non-evaluable due to only 4 weeks of ANP and lack of follow-up scans. One patient who had stable disease discontinued ANP against medical advice and died 4.5 years later. Ten patients are alive and well from 2 to >14 years post-diagnosis. Only one case of serious toxicity of reversible tinnitus, of one day’s duration, was described. The study continues with accrual of additional patients.
The results of the present study are favourable in comparison with radiation therapy and chemotherapy. We believe that confirmation of these results through further studies may introduce a new promising treatment for incurable paediatric brain tumours.”
“The patients were diagnosed by pathologists not associated with BC and objective responses were verified by Central Radiology Review. The group of 77 patients (19%) survived over five years from the treatment start. Of particular interest were results in patients with brainstem gliomas. The group of 17 patients with brainstem glioma underwent the treatment and 65% of these patients survived over five years. An additional group of 42 patients diagnosed with diffuse intrinsic pontine glioma (DIPG) have been treated and a total of 19% survived over five years.
The quality of survival is very good and there is no long-term toxicity related to ANP…”





